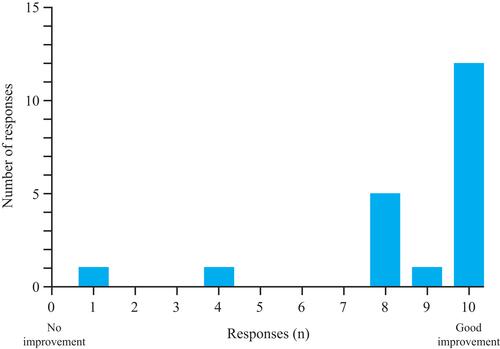
Figures & data
Figure 1 Design of the retrospective study.
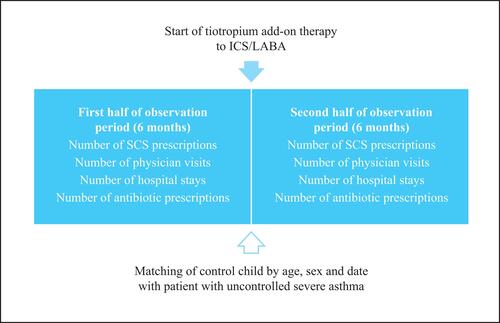
Table 1 Patient and Control Demographics Before Switching to ICS/LABA/Tiotropium Treatment
Figure 2 (A) Systemic corticosteroid administration; (B) physician visits; (C) hospitalisations; (D) use of antibiotics. Data are shown as mean±standard error of mean. Pre=observation period 1 (up to 6 months before start of ICS/LABA/tiotropium therapy); post=observation period 2 (up to 6 months after start of ICS/LABA/tiotropium therapy). Treatment differences of tiotropium, physician visits and antibiotic use between pre and post observation were evaluated by the Wilcoxon–Mann–Whitney test. Abbreviations: ICS, inhaled corticosteroids; LABA, long-acting β2-agonist.
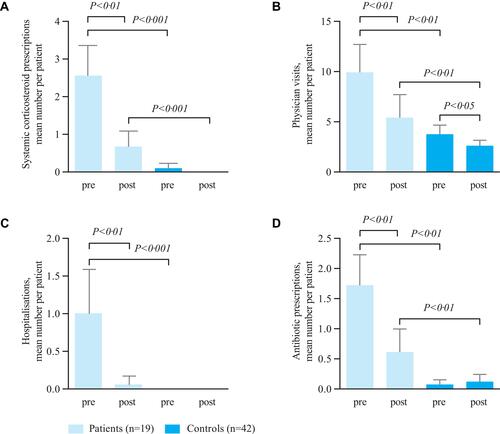
Table 2 Treatment Response in Patients with and without LTRA Use