Figures & data
Figure 1 Model of GR transactivation and transrepression of genes, when induced by classical GCs and SEGRAs. SEGRAs have been designed to improve the therapeutic index compared to classical GCs, by preferentially inducing transrepression with negligible transactivating activity.
Abbreviations: GC, glucocorticoid; GR, glucocorticoid receptor; GRE, glucocorticoid response element; SEGRA, selective glucocorticoid receptor agonist; TF, transcription factor; TFRE, transcription factor responsive element.
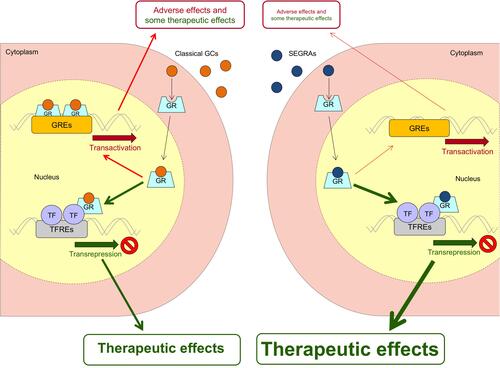
Figure 2 PRISMA flow diagram for the identification of the studies included in the systematic review concerning the impact of experimental GR agonists for the treatment of asthma.
Abbreviations: GC, glucocorticoid; GR, glucocorticoid receptor; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
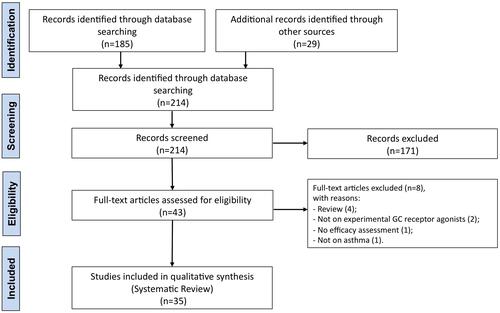
Table 1 Characteristics of the Clinical Studies Included in the Systematic Review
Figure 3 Weighted plot for the assessment of the overall risk of bias via the Cochrane RoB 2 tool (n=15 RCTs).
Abbreviation: RCT, randomized controlled trial.
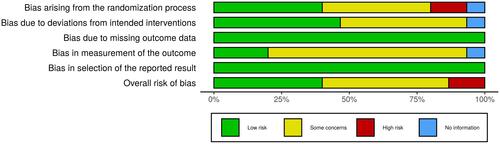
Figure 4 Traffic light plot for assessing the specific risk of bias of each RCT via the Cochrane RoB 2 tool. D1: bias arising from the randomization process; D2: bias due to deviations from intended intervention; D3: bias due to missing outcome data; D4: bias in measurement of the outcome; D5: bias in selection of the reported result. Red circle indicates high risk of bias, yellow circle indicates some concerns on the risk of bias, green circle represents low risk of bias, and blue circle refers to lack of information due to limited data results available on Clinicaltrials.gov database.
Abbreviations: RCT, randomized controlled trial; RoB, Risk of Bias.
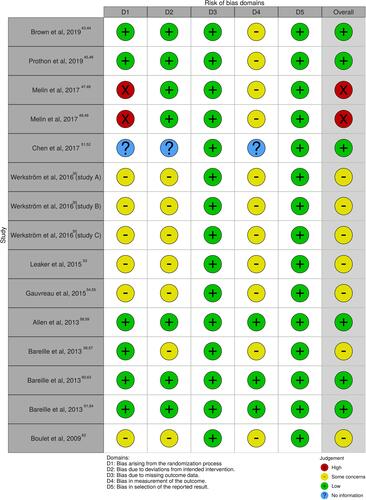
Table 2 Main PK Characteristics of Investigational GR Agonists for the Treatment of Asthma
