Figures & data
Figure 1 Fluorescent inhibitors 1, 2 used to validate hCA IX/XII as antitumor drug targetsCitation60–Citation63 and the pan-inhibitor acetazolamide 3.
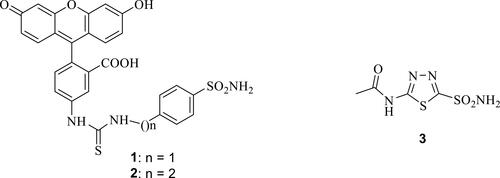
Figure 2 Acetazolamide-based CAIs 4, 5 and 7 reported by Neri’s group and the cytotoxin 6 (DM1).Citation64,Citation66
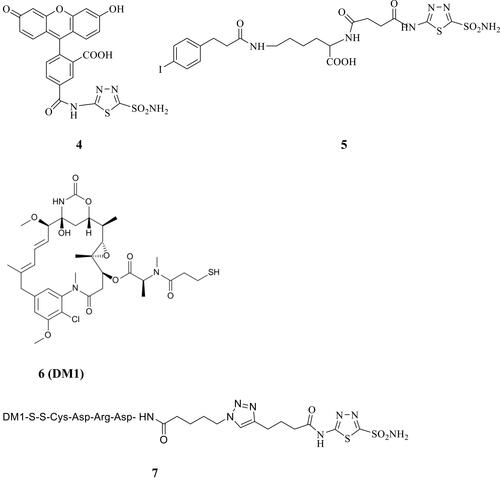
Figure 3 Nitroazole-containing CAIs of types 8–11.Citation74–Citation79
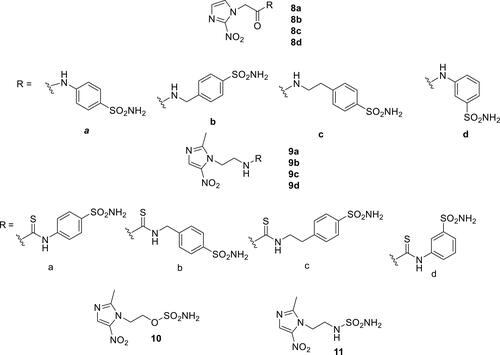
Figure 4 Sulfamates incorporating ureido (compounds 12, 14 and 15) and thioureido (13) moieties, with effective CA IX/XII inhibitory action and investigated for their antitumor effects. S4 was the most extensively investigated such derivative.Citation80
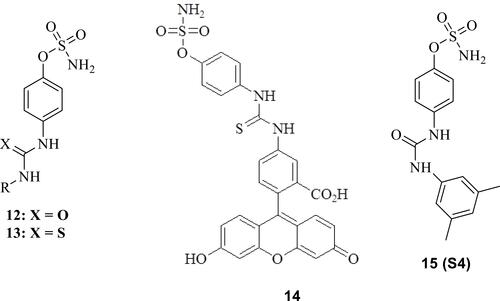
Figure 5 Examples of glycomimetic CAIs incorporating benzenesulfonamide, sugar and 1,2,3-triazole moieties, of types 16–22.Citation82–Citation89
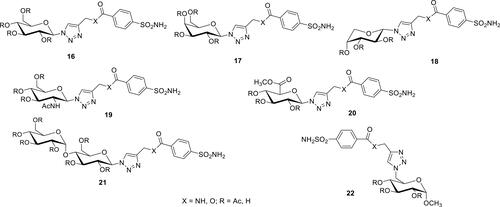
Figure 6 The natural product sulfonamide Psammaplin C, 23.Citation96
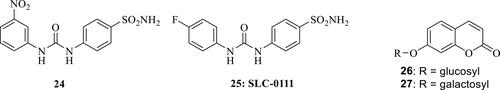
Figure 7 Ureido-sulfonamides 24 and 25 and 7-glycosyl-substituted coumarins 26, 27.Citation98–Citation101