Figures & data
Figure 1 Glycolytic activity is enhanced in FLS of RA patients. Up-regulation of HK2 enhances the migration and invasion ability of FLS, which serves as an example how glycolysis drives RA. Many glycolytic intermediates like succinate up-regulate glycolysis-related genes via HIF-1α, further facilitate this metabolic process. Above changes eventually fuel vascular proliferation, inflammation, and cartilage damages. HIF-1α plays a key role in related signal transduction and pathological changes.
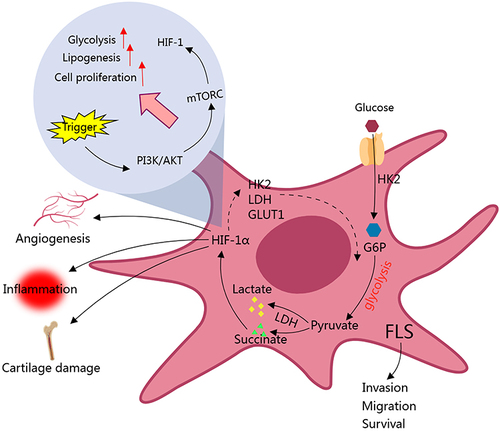
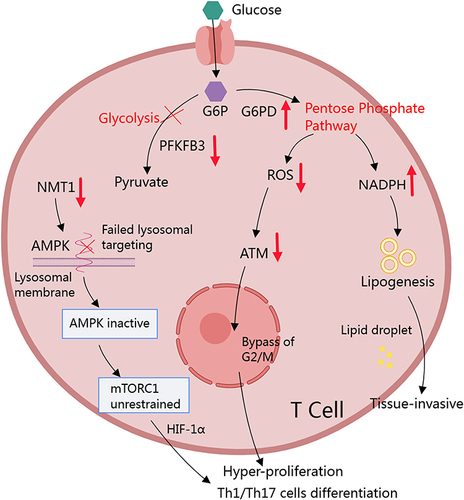
Figure 2 RA conditions favor PPP in CD4+ T cells. Increased PFKFB3/G6PD ratio serves as the fundament for the accelerated PPP in RA T cells. It leads to the production of large amounts of NADPH in cytoplasm. By supporting lipid synthesis, NADPH facilitates membranes construction, which makes T cells more reproductive and invasive. The increase in NADPH eventually depletes ROS. It directly prolongs the longevity of T cells, and induces them to rapidly proliferate by bypassing ATM-controlled G2/M cell cycle checkpoint. The low oxidative stress condition is also favorable for Th1 and Th17 differentiation. Concurrently, decreased NMT1 expression in T cells results in the inability to target AMPK to lysosomes, leading to elevated and unrestricted activation of mTORC1, which promotes Th1 and Th17 differentiation by mediating HIF-1α.