Figures & data
Figure 1 Signaling of IL-36, IL-37 and IL-38. IL-36α, IL-36β and IL-36γ counteract with IL-36Ra, IL-38, and then bind to IL-36R, IL-1RAcP. Moreover, activating signalings were transported to intracellular cascades, including IKK, which will activate signalings such as IκB, NF-κB, PI3K/AKT, ERK. Subsequently, transcription factors mTORC, WNT5A in nucleus will regulate inflammatory cytokines, chemokines, growth factors production. IL-37 binds to receptors IL-1R8, IL-18Rɑ, and then activate signalings such as PI3K/ATK, ERK, JNK, P38. Activated cascades will regulate Notch1, NRF2 activation by STAT3, SIGIRR. Finally, expression of pro-inflammatory or anti-inflammatory components in nucleus were regulated by the IL-37 axis. IL-38 binds to receptors IL-36R or IL-1RAPL1 by competing with the IL-36α/β/γ. Then, the activating signalings were transmitted to cascades RohA, ERK, JNK, P38, and subsequently, transcription factors AP-1, SIRT1 were activated. Finally, inflammatory factors in nucleus were regulated by the IL-38 axis.
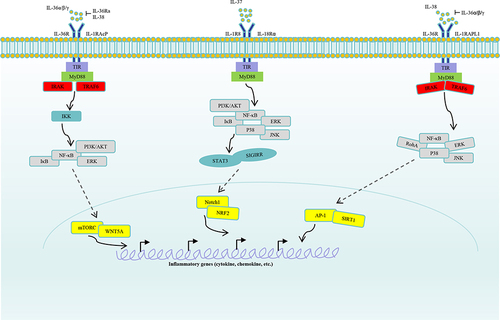
Figure 2 Function of IL-37 in different immune cells. IL-37 inhibits expression of inflammatory components such as iNOS, IL-6, MCP-1, ROS in macrophages, and promotes expression of IL-10, GPX4, NRF2, CD206. IL-37 inhibits dendritic cells maturation, including downregulation of expression of MHCII, CD40, CD86, CD80. IL-37 suppresses infiltration of basophils, and Th17, Tfh cells differentiation, proliferation, whereas it promotes Th1, Treg cells differentiation. IL-37 is able to inhibit B cells production of IgG.
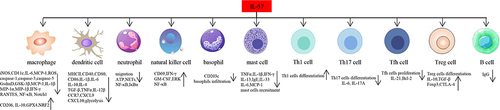
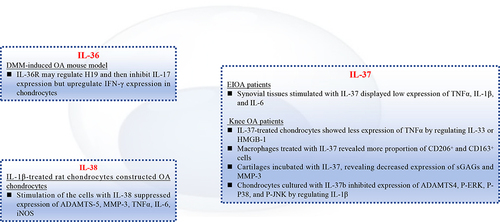
Table 1 Expression of IL-36, IL-37 and IL-38 in Osteoarthritis and Rheumatoid Arthritis
Figure 3 In vivo effects of IL-36, IL-37 and IL-38 on OA development. Injection of IL-36Ra inhibited Tgfbr2−/− spontaneous OA mice synovitis, whereas injection of IL-36α promoted OA development. In DMM-induced OA mice, injection of IL-36Ra or IL-36Ra@Gel suppressed severity of cartilage tissue destruction. On the contrary, injection of IL-36α exacerbated OA progress. For IL-37, injection of IL-37 into TMJOA rats suppressed swelling and hyperplasia in tissues around the cartilage. Regarding role of IL-38, DMM-induced OA mice injected with overexpressed IL-38 significantly improved cartilage damage, downregulated OARSI score.
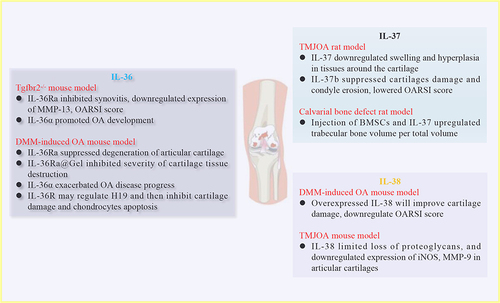
Figure 4 In vitro role of the IL-1 family cytokines in OA chondrocytes and macrophages. IL-36R may regulate H19, then, inhibit expression of IL-17 in chondrocytes from DMM-induced OA mice. Synovial tissues from EIOA patients stimulated with IL-37 will reveal low expression of TNFα, IL-1β, and IL-6. Macrophages from knee OA patients were treated with IL-37, showing more CD206+ and CD163+ cells. IL-1β-treated rat chondrocytes constructed OA chondrocytes, evidenced by high expression of ADAMTS-5, MMP-3, TNFα. However, addition of IL-38 suppressed the pro-inflammatory cytokines.