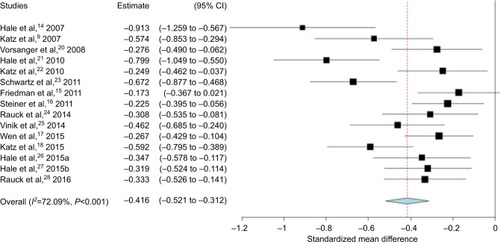
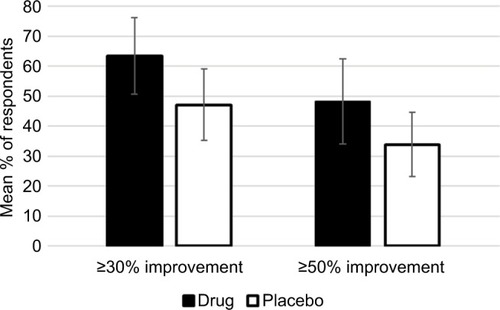
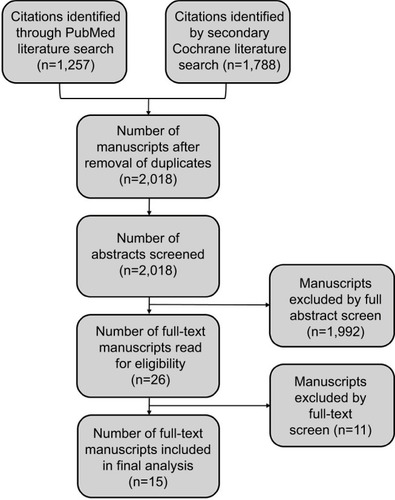
Figures & data
Table 1 Study design and subject disposition by study
Table 2 Summary of secondary endpoints