Figures & data
Table 1 Key inclusion and exclusion criteria
Figure 1 Study flow chart and patient profile.
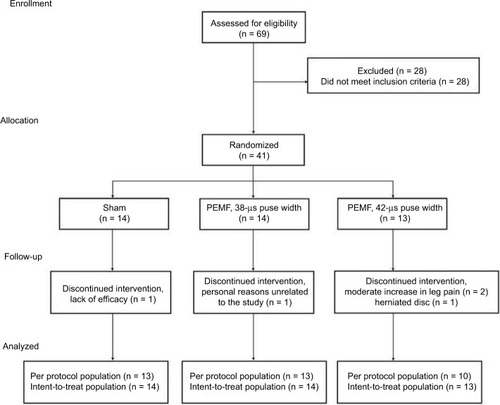
Table 2 Subject demographics and baseline characteristics
Figure 2 Mean pain scores at the end of treatment (Days 56–60) compared with baseline.
Abbreviation: NPRS, numerical pain rating scale.
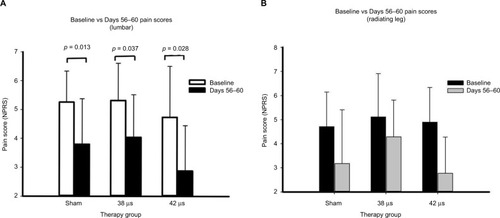
Figure 3 Mean pain scores over time.
Abbreviations: NPRS, numerical pain rating scale; PEMF, pulsed electromagnetic field.
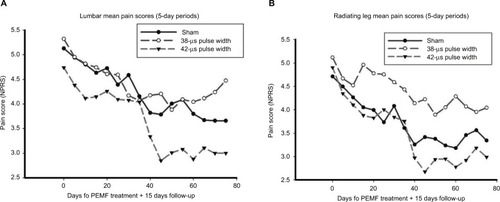
Figure 4 Mean pain scores over time normalized to baseline.
Abbreviation: PEMF, pulsed electromagnetic field.
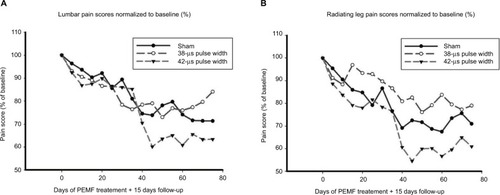
Table 3 Primary outcome measure at 60 days
Table 4 Secondary outcome measures at 60 days
Figure 5 Time to 30% reduction in pain score is the time to response. Figures show the time to 30% reduction, which was evaluated using Kaplan–Meier statistics.
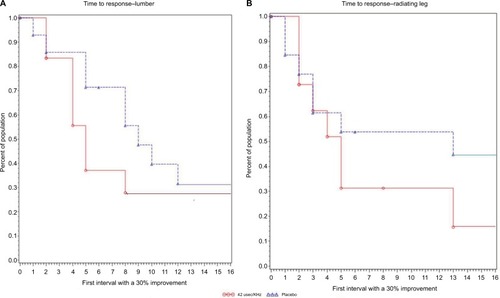
Table S1 Titles of participating sites:
