Figures & data
Figure 1 Neonatal repeated sevoflurane exposure enhances the sensitivity to pain later in juvenile life, Pretreatment with bumetanide prior to neonatal sevoflurane exposure alleviated this response. PWTL: The paw withdrawal thermal latency. (mean ± SEM; n = 10 rats/group; *p < 0.05 vs CON group; #p < 0.05 vs SEV group, one-way ANOVA).
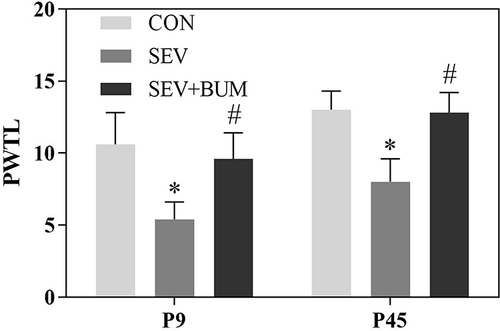
Figure 2 Neonatal repeated exposure to sevoflurane in rats enhanced the secretion of corticosterone in the juvenile CFTS model, while the basal secretion of serum corticosterone was not significantly affected at juvenile period (mean ± SEM; n = 6 rats/group; *p < 0.05 vs CON group; #p < 0.05 vs SEV group, one-way ANOVA).
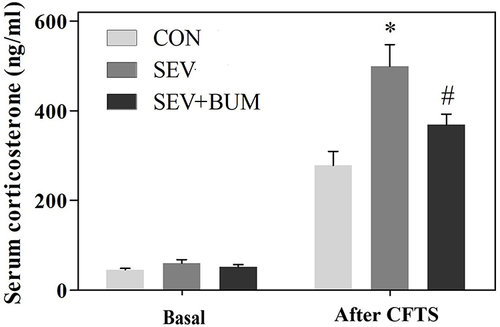
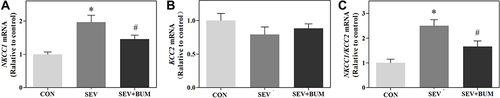
Figure 3 Repeated sevoflurane exposures in neonatal rats at P6, P7, P8 increases the NKCC1/KCC2 mRNA ratio in the hypothalamus of rat pups, which was alleviated by pretreated with the NKCC1 inhibitor bumetanide. (A) Repeated sevoflurane exposures in neonatal rats at P6, P7, P8 increased the expression of NKCC1 mRNA in the hypothalamus. (B) Repeated sevoflurane exposures in neonatal rats at P6, P7, P8 had no significant effects on expression of KCC2 mRNA in the hypothalamus. (C) Repeated sevoflurane exposures in neonatal rats at P6, P7, P8 increases the NKCC1/KCC2 mRNA ratio in the hypothalamus of rat pups. (mean ± SEM; n = 6 rats/group; *p < 0.05 vs CON group; #p < 0.05 vs SEV group, one-way ANOVA).