Figures & data
Table 1 Summary of baseline patient characteristics and medication use
Table 2 Patient level data grouped by those who achieved ≥50% decrease in PANSS negative score from baseline to week 8 and those who did not
Table 3 Summary of treatment effectiveness in patients with predominantly negative symptoms of schizophrenia and corresponding average week 8 dose of amisulpride
Figure 1 Summary of changes in PANSS total score, and positive and negative symptom scores over 8 weeks of amisulpride treatment in patients with predominantly negative symptoms of schizophrenia.
Abbreviation: PANSS, Positive and Negative Syndrome Scale.
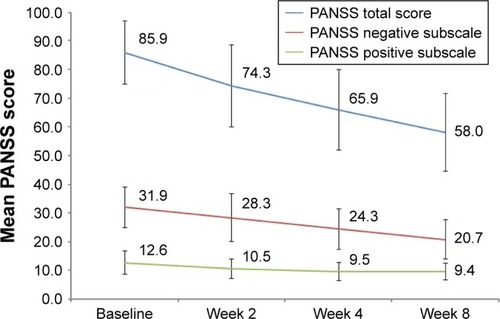
Figure 2 A comparison of the mean decrease in PANSS negative and total score and CGI-S score from baseline to weeks 2, 4 and 8 in patients with predominantly negative symptoms of schizophrenia.
Abbreviations: CGI-S, Global Clinical Impression Severity Scale; PANSS, Positive and Negative Syndrome Scale.
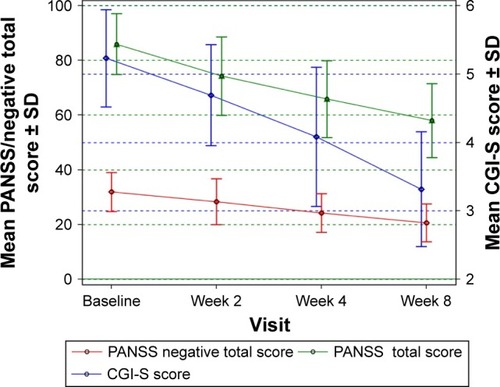
Figure 3 Change in individual items of the PANSS negative symptom score from baseline to weeks 2, 4 and 8 in patients with predominantly negative symptoms of schizophrenia.
Abbreviation: PANSS, Positive and Negative Syndrome Scale.
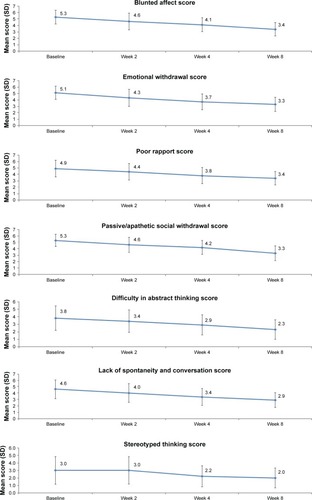
Table 4 Summary of safety data
