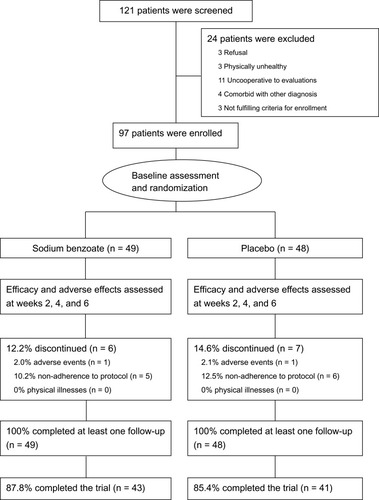
Figures & data
Table 1 Results of Measures of Serum DAAO, Plasma Amino Acids, and Plasma Antioxidants Over the 6-Week Treatment Between Sodium Benzoate and Placebo Groups
Table 2 Zero-Order Correlations Among Laboratory and Clinical Measures at Baseline and Their Difference Values (Endpoint – Baseline)
Table 3 Multiple Linear Regression Analyses of Independent Factors Associated with ADAS-Cog and BEHAVE-AD Improvements (Score Reduction from Baseline to Endpoint) in Sodium Benzoate Group (Stepwise)
Table 4 Multiple Linear Regression Analyses of Independent Factors Associated with ADAS-Cog and BEHAVE-AD Improvements (Score Reduction from Baseline to Endpoint) in Placebo Group (Stepwise)
Data Availability Statement
The protocol of this study is online (https://clinicaltrials.gov/ct2/show/NCT02103673). The individual de-identified participant data are available to qualified investigators on request to the corresponding author after the request is approved by the Institutional Review Board and Data Safety Monitoring Board of Kaohsiung Chang Gung Memorial Hospital, Institutional Review Board and Data Safety Monitoring Board of China Medical University Hospital, and Institutional Review Board and Data Safety Monitoring Board of Lin-Shin Hospital.