Figures & data
Table 1 Measures at study entry for patients whose antipsychotic medication was Switched (n = 53) or Augmented (n = 34) during the 12-month study period
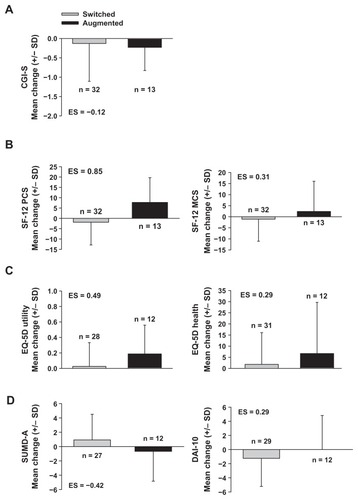
Figure 1 Effect sizes and changes from study entry to time of first switch or augmentation among patients experiencing a switch to (gray bars), or augmentation with (black bars), another oral antipsychotic following study initiation: (A) illness severity, as measured by the CGI-S, (B) functioning and well-being, as measured by the SF-12 physical (left) and mental component score (right), (C) quality of life, as measured by the EQ-5D utility index (left) and health state score (right), and (D) attitude towards medication intake and insight into illness, as measured by the SUMD-A (left) and DAI-10 (right), respectively.
Abbreviations: CGI-S, Clinical Global Impressions-Severity scale; DAI-10, 10-item Drug Attitude Inventory; EQ-5D, 5-dimension European Quality of Life scale; MCS, mental health component score; ES, effect size for Augmented patient group, relative to Switched patient group, on change scores from study entry to time of first switch/augmentation event; PCS, physical component score; SF-12, 12-item Short-Form Health Survey; SD, standard deviation; SUMD-A, Scale to Assess Unawareness of Mental Disorder-Abbreviated version.