Figures & data
Table 1 Selection criteria of systematic review
Figure 1 PRISMA diagram of the literature selection criteria.
Abbreviations: PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; ADHD, attention-deficit/hyperactivity disorder.
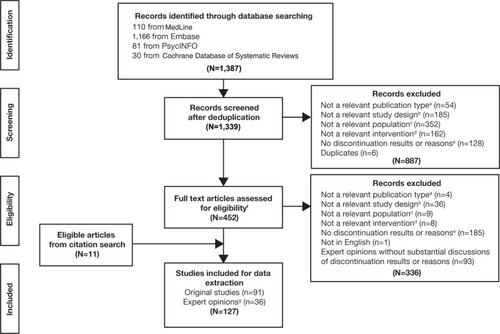
Table 2 Number of studies reporting results for subgroups
Table 3 Studies included in quantitative analysis for persistence
Table 4 Persistence results (reported as mean treatment duration, in days) from quantitative analysis
Table 5 Studies included in quantitative analysis for adherence
Table 6 Adherence results (mean MPR) from quantitative analysis
Table 7 Most frequently reported reasons for treatment discontinuation in original studies
Table 8 Most frequently reported reasons for treatment discontinuation in expert opinions
Table S1 A list of ADHD medications approved in North American and European countries
Table S2 List of included original observational studies and bibliographic information
Table S3 List of included studies with expert opinions and bibliographic information
