Figures & data
Table 1 Basic Demographic Information of the Participants (n=88)
Figure 1 Understanding of “Clinical Trials” and “Clinical Trial Participants” among 88 Participants.
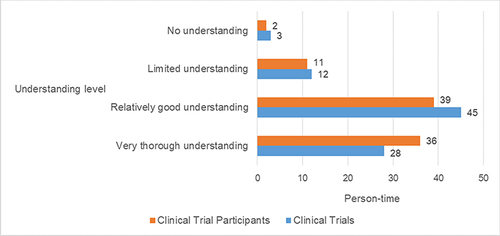
Table 2 Levels of Understanding of the Concept of “Clinical Trials” and “Clinical Trial Participants” Among 88 Participants with Differing Educational Backgrounds
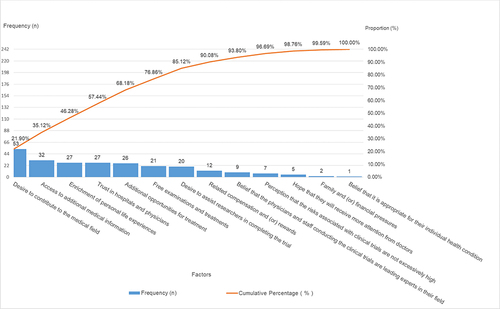
Table 3 Factors Influencing Participants’ Willingness to Partake in Clinical Trials: Category a (Major Factors), Category B (Minor Factors), and Category C (General Factors)
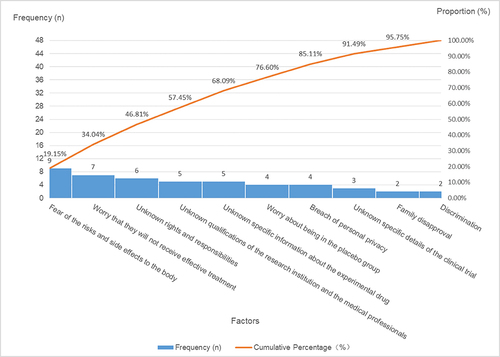
Table 4 Factors Influencing Participants’ Hesitation to Partake in Clinical Trials: Category a (Major Factors), Category B (Minor Factors), and Category C (General Factors)
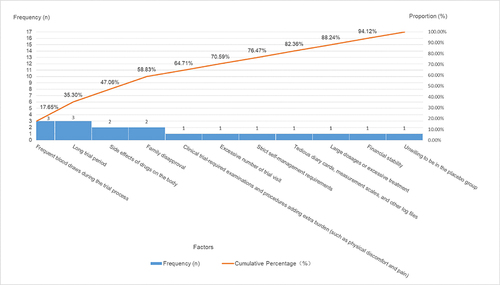
Table 5 Factors Influencing Participants’ Unwillingness to Partake in Clinical Trials: Category a (Major Factors), Category B (Minor Factors), and Category C (General Factors)
Table 6 Confidence Levels Regarding the Efficacy of Experimental Drugs Among Participants with Different Occupations
Table 7 Willingness Level to Participate in Clinical Trials Among Different Population Groups Based on Their Level of Confidence in the Effectiveness of Drugs Trials
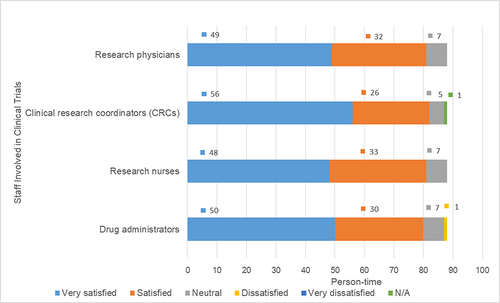
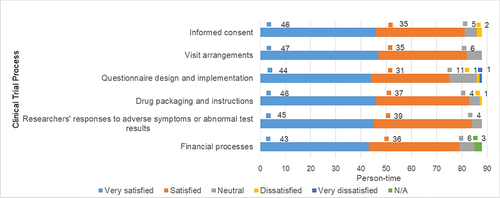
Table 8 Overall Satisfaction Ratings of 88 Participants in Clinical Trials
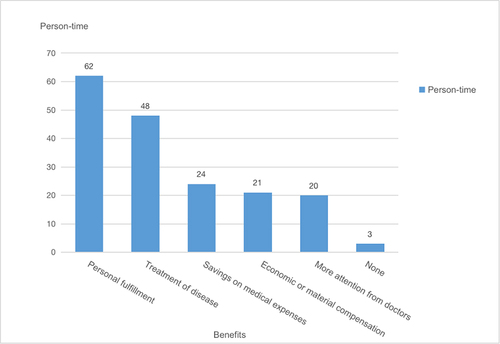
Table 9 Additional Unmet Needs
Table 10 Recommendations for Enhancing Satisfaction Among Clinical Trial Participants
Data Sharing Statement
The data that support the findings of this study may be provided by the corresponding author upon reasonable request.