Figures & data
Figure 1 Allergan’s handheld TrueTear® intranasal tear neurostimulator device features two prongs covered in disposable hydrogel tips. The tips are inserted into the nasal cavity and provide electrical stimulation to the trigeminal nerve. Adapted from US Food & Drug Administration. De Novo Classification Request for Intranasal Tear Neurostimulator. Available from: https://www.accessdata.fda.gov/cdrh_docs/reviews/DEN160030.pdf.Citation55
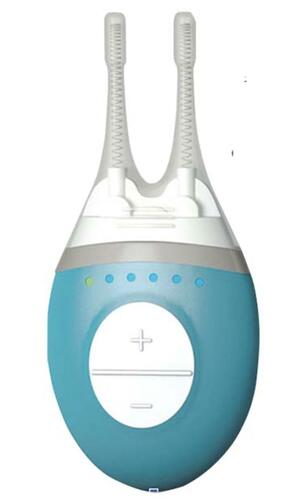
Figure 2 The commercial iTEAR100 device applies repetitive electromechanical stimulation to the external nasal nerve via an oscillating tip (black arrow). Reproduced from Ji MH, Moshfeghi DM, Periman L, et al. Novel Extranasal Tear Stimulation: Pivotal Study Results. Transl Vis Sci Technol. 2020;9(12):23. Available from: https://tvst.arvojournals.org/article.aspx?articleid=2771969&resultClick=1.Citation32

