Figures & data
Table 1 Demographics and previous ocular topical therapies of the study population
Figure 1 Improvement on the subscale scores of the Impact on Dry Eye in Everyday Life questionnaire, from baseline (with habitual drops) to visit 4 (after 4 weeks of treatment with study drops). (A) Mean change per patient; maximum possible change was 100 units. (B) Clinical significance of improvement (effect size indices).Notes: Error bars represent the standard deviation and are shown unidirectional for clarity. *P < 0.01; **P < 0.001.
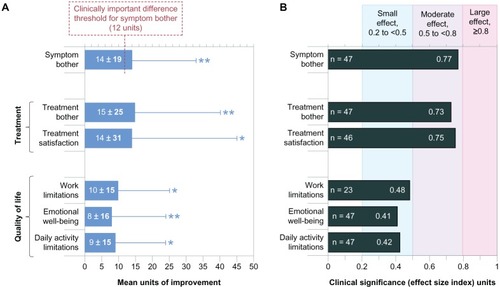
Table 2 Scores on the subscales of the Impact on Dry Eye in Everyday Life questionnaire at baseline (with habitual drops) and at visit 4 (after 4 weeks of treatment with study drops)
Table 3 Results of the Work Productivity and Activity Impairment Questionnaire analyses
Table 4 Patient assessments of study medication after 4 weeks of treatment
Table 5 Patient satisfaction with study medication after 4 weeks of treatment
Figure 2 Dosing frequency for the 46 study participants whose electronic dosage records were complete at both time points (after 1 week of habitual therapy and after 4 weeks of study medication).
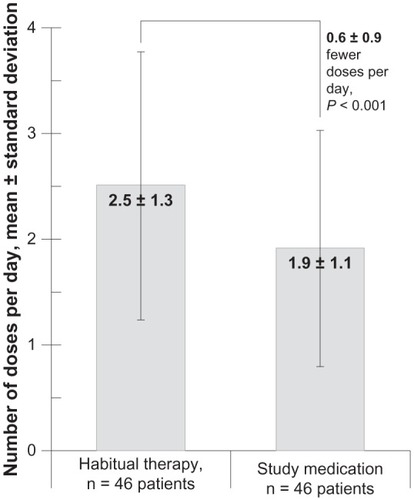
Table 6 Tear film breakup time
Table 7 Corneal staining
Table 8 Comparison of signs of meibomian gland dysfunction in two studies of patients treated with topical cyclosporine, artificial tears, or Systane® Balance Lubricant Eye Drops (Alcon Inc, Fort Worth, TX, USA)