Figures & data
Table 1 Patient demographics
Table 2 Change in mean intraocular pressure (IOP) from baseline to final visit 12 weeks after initiation of medical therapy with preservative-free tafluprost
Figure 1 Percentage intraocular pressure (IOP) reduction at final visit versus untreated baseline for different patient subgroups.
Abbreviations: NTG, normal tension glaucoma; OH, ocular hypertension; PEX, exfoliative glaucoma; POAG, primary open-angle glaucoma.
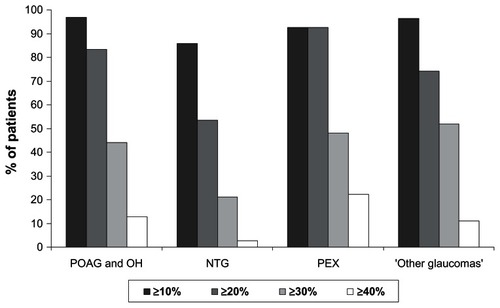
Figure 2 Achievement of specific intraocular pressure levels at final visit versus untreated baseline for different patient subgroups.
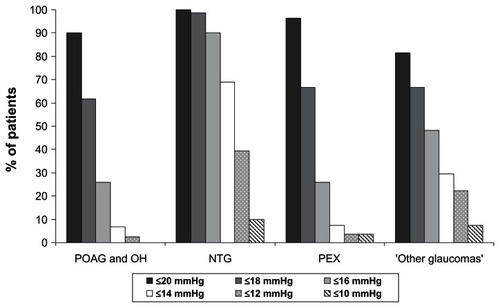
Figure 3 Achievement of specific intraocular pressure (IOP) levels at final visit versus untreated baseline in all patients with primary open-angle glaucoma (POAG) and ocular hypertension (OH) and in the two subgroups stratified by baseline IOP levels at month 3 after initiation of medical treatment.
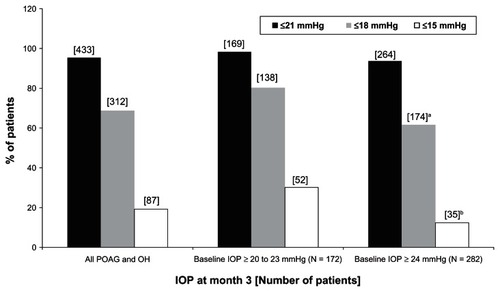
Table 3 Intraocular pressure (IOP) ± standard deviation (SD) in patients with primary open-angle glaucoma and ocular hypertension at baseline and at month 3 after initiation of medical treatment
Figure 4 Local tolerability of preservative-free tafluprost at month 3 for all patients and for different patient subgroups.
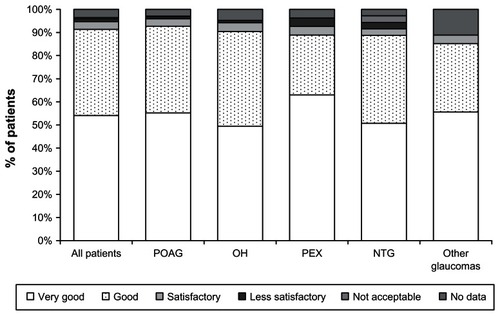
Table 4 Adverse events and terminations of treatment