Figures & data
Table 1 Patient characteristics and methodologic summary of four open studies of Hylabak® in patients with dry eye syndrome
Table 2 Etiology of dry eye syndrome in each study
Figure 1 Change in subjective discomfort score during 28 days of treatment with Hylabak® in (A) postmenopausal women, (B) patients with persistent meibomian blepharitis, and (C) Sjögren’s syndrome (n=32)Citation25 compared with a similar group of patients treated with Tear Naturale® (n=25).Citation28
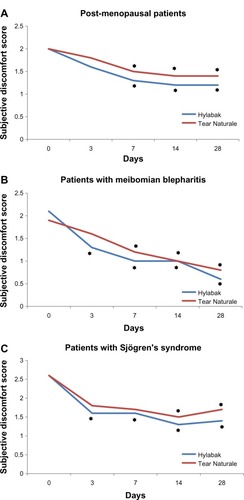
Figure 2 Change in objective signs score during 28 days of treatment with Hylabak® in (A) postmenopausal women, (B) patients with persistent meibomian blepharitis, and (C) Sjögren’s syndrome (n=32)Citation25 compared with a similar group of patients treated with Tear Naturale® (n=25).Citation28
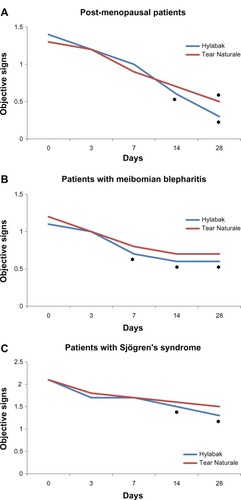
Figure 3 Change in subjective signs and symptoms score during 2 weeks of treatment with Hylabak® in office workers with (A) mild or (B) moderate symptoms (n=32).Citation26
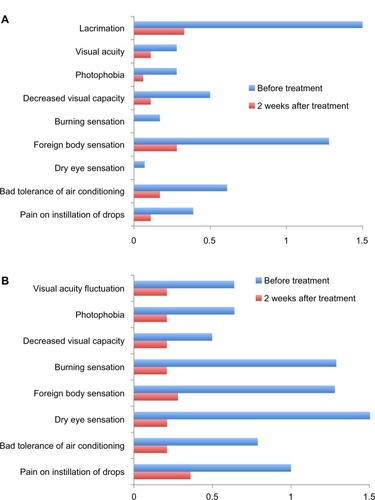
Figure 4 Change in number of patients with subjective complaints during 2 months of treatment with Hylabak® in children and adolescents wearing soft contact lenses (n=20).Citation27
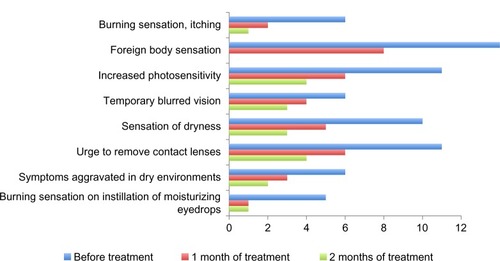
Table 3 Results of functional tests (Schirmer’s test and Norn’s test) performed in all four studies
