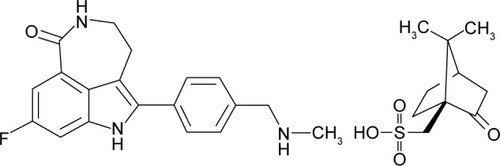
Figures & data
Table 1 Pharmacokinetics of rucaparib
Figure 2 ARIEL3 schema.
Notes: N=540; primary end point: PFS; secondary end points: OS, health-related quality of life, and safety and tolerability; analysis will evaluate the three separate groups defined in ARIEL2. Clovis Oncology. A Study of Rucaparib as Switch Maintenance Following Platinum-Based Chemotherapy in Patients With Platinum-Sensitive, High-Grade Serous or Endometrioid Epithelial Ovarian, Primary Peritoneal or Fallopian Tube Cancer (ARIEL3). Available from https://clinicaltrials.gov/ct2/show/NCT01968213?term=NCT01968213&rank=1. NLM identifier: NCT01968213.Citation48
Abbreviations: b.i.d., twice daily; CR, complete response; PFS, progression-free survival; p.o., by mouth; PR, partial response; OS, overall survival; R, randomized.
Abbreviations: b.i.d., twice daily; CR, complete response; PFS, progression-free survival; p.o., by mouth; PR, partial response; OS, overall survival; R, randomized.
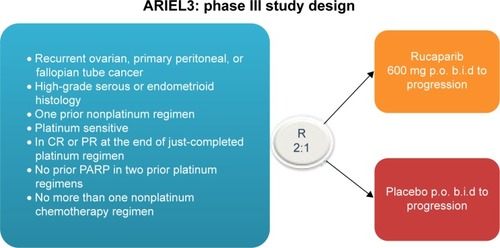
Table 2 Rucaparib in epithelial ovarian cancer clinical trials