Figures & data
Table 1 Patient clinical information
Figure 1 Adverse effects of the two treatment.
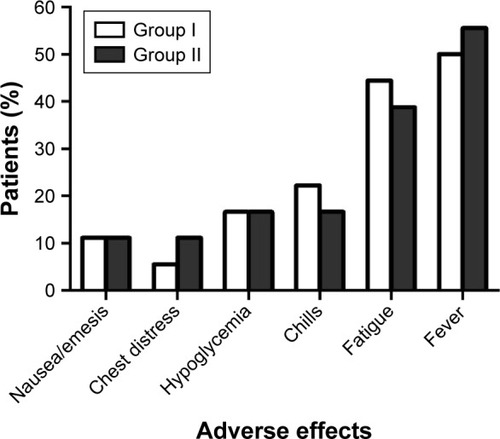
Table 2 Comparison of immune function between the two groups
Figure 2 Changes in the CTC level before treatment and 2 months after treatment.
Abbreviation: CTC, circulating tumor cell.
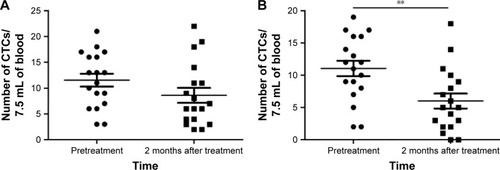
Figure 3 Flow cytometry images of a 53-year-old patient in group II.
Abbreviations: APC, allophycocyan; CTC, circulating tumor cell; FITC, fluorescein isothiocyanate; PE, phycoerythrin.
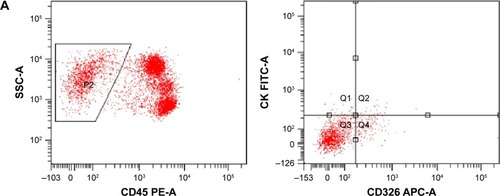
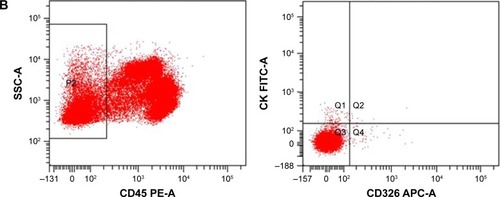
Figure 4 CT images of a 46-year-old patient with breast cancer.
Abbreviation: CT, computed tomography.
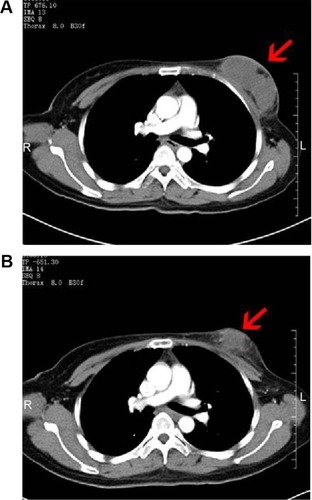
Table 3 Maximum transverse diameter and CT value of the lesions before and after treatment
Table 4 Comparison of the curative effect between the two groups at 2 months post-treatment
Figure 5 Changes in the KPS.
Abbreviation: KPS, Karnofsky performance status.
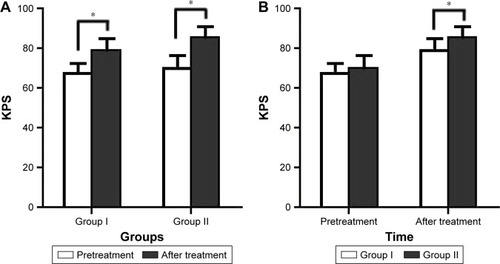
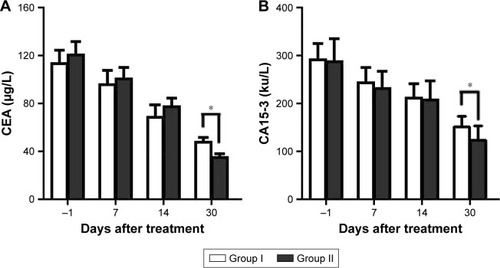
Figure 6 Changes in the biochemical indicators.
Abbreviations: CA15-3, cancer antigen 15-3; CEA, carcinoembryonic antigen.