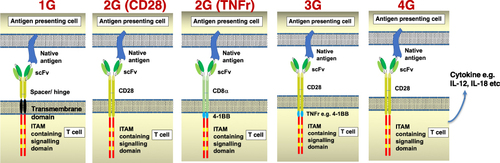
Figures & data
Table 1 Clinical Trials Evaluating CAR T-Cell Immunotherapy of Lung Cancer