Figures & data
Table 1 Clinical and pathologic characteristics of the patients
Figure 1 Immunostaining for TOP2A in preoperative breast cancer core biopsies.
Abbreviation: TOP2A, topoisomerase II alpha.
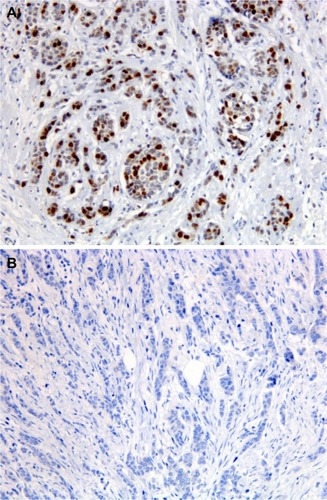
Figure 2 Immunostaining for TLE3 in preoperative breast cancer core biopsies.
Abbreviation: TLE3, transducin-like enhancer of split 3.
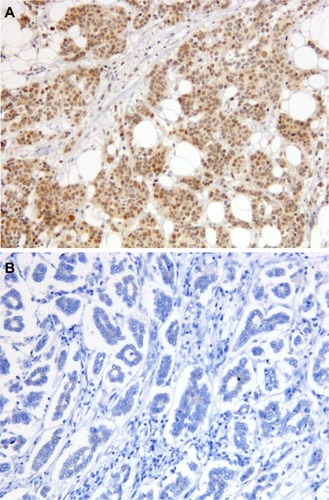
Table 2 Tumor diameter before and after neoadjuvant chemotherapy treatment
Table 3 Distribution by size reduction (%) after chemotherapy treatment
Table 4 Probability to obtain a tumor size reduction ≥70% after neoadjuvant chemotherapy according to clinical–pathological and IHC features (logistic regression analysis)
Figure 3 Distribution of good responders according toTOP2A status.
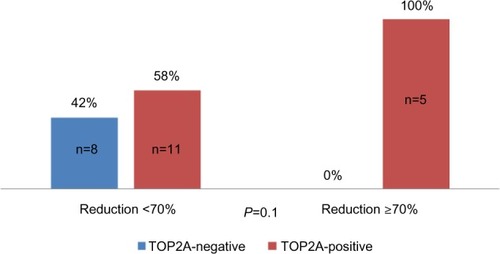
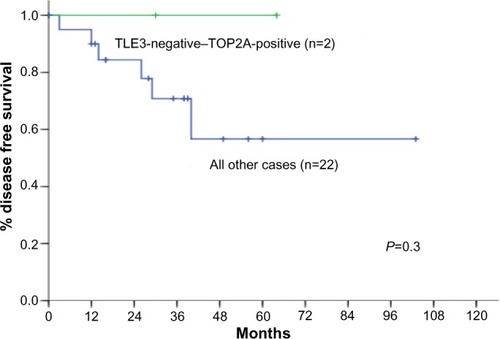
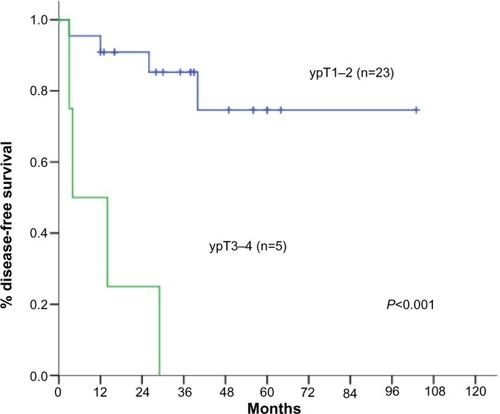
Figure 4 Disease-free survival according to TOP2A positivity.
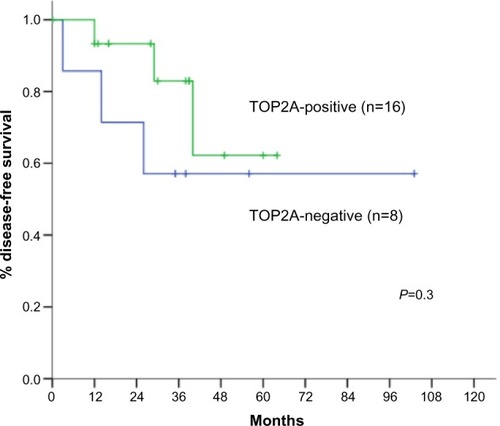
Figure 5 Distribution of good responders according to TLE3 status.
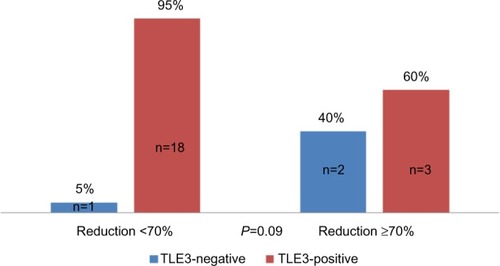
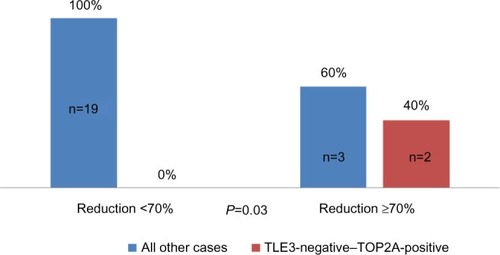
Figure 6 Distribution of good responders according to concurrent TOP2A positivity and TLE3 negativity.