Figures & data
Table 1 Attributes and levels included in the scenarios
Table 2 Patient sociodemographic and clinical variables
Table 3 Number and percentage of patients receiving treatment with BAs at the time of study inclusion, and previous treatment
Figure 1 Patients’ (A) and rheumatologists’ (B) utility values.
Abbreviation: AEs, adverse events.
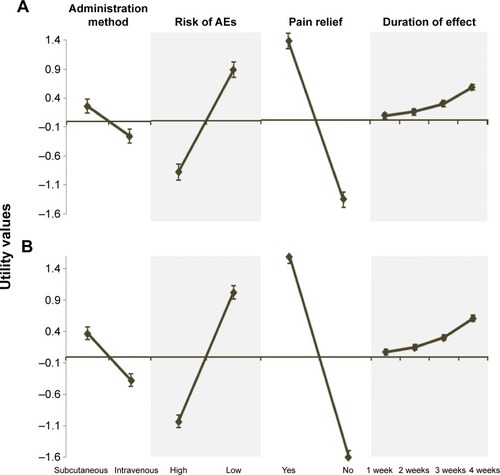
Figure 2 Relative importance values given by both patients and rheumatologists to the attributes of biological agents and for specific rheumatic conditions.
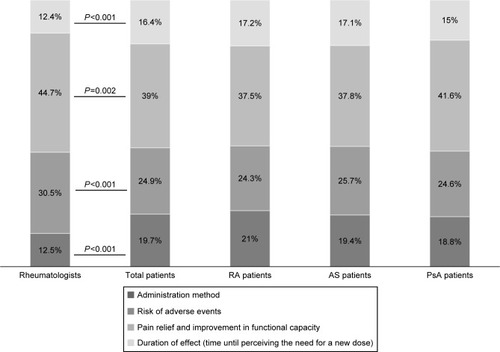
Figure 3 Pareto diagrams representing patients’ and professionals’ utility values for subcutaneous and intravenous treatment alternatives.
Abbreviations: AEs, adverse events; BA, biological agent.
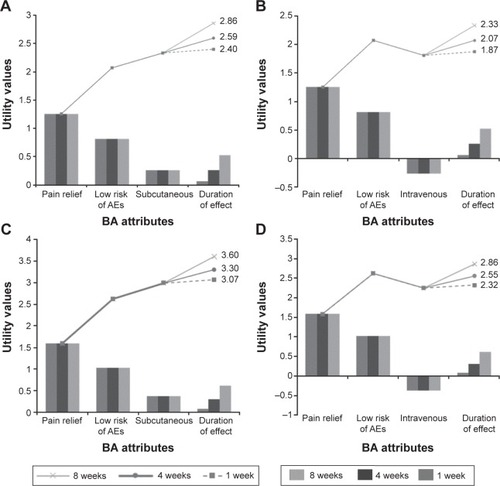
Table 4 Factors influencing patients’ and rheumatologists’ preferencesTable Footnotea
