Figures & data
Figure 1 Study design of Study CGAJ.
Abbreviation: SP, study period.
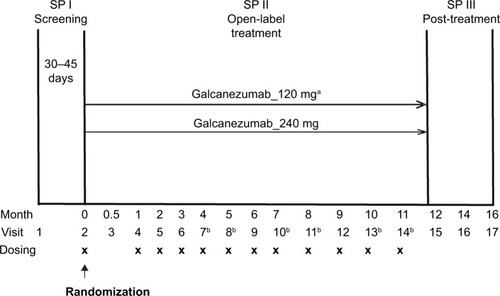
Table 1 Baseline demographics and clinical characteristics
Figure 2 Reduction in number of migraine headache days per month in patients with episodic migraine (A) and chronic migraine (B).
Abbreviations: GMB, galcanezumab; LS, least square; SE, standard error.
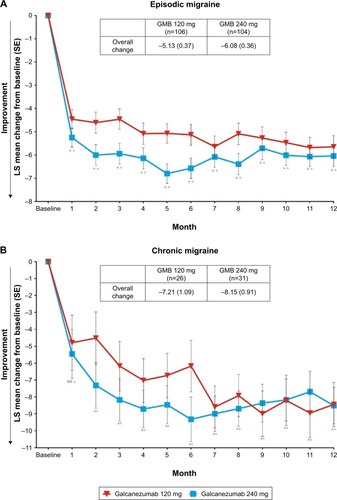
Table 2 Percentage of patients in the combined galcanezumab dose groups with positive responses on the Patient Satisfaction with Medication Questionnaire–Modified
Figure 3 Migraine-specific HCRU as count of persons with at least one visit (%) (A), and as per 100 patient-years (B) during baseline period and treatment period.
Abbreviations: ER, emergency room; GMB, galcanezumab; HCP, health care professional.
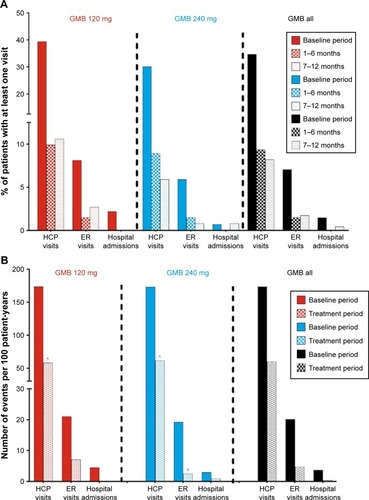
Figure 4 Change from baseline in number of days per month with acute medication use for migraine or headache. Changes at each month (A) and overall change (B) from baseline are shown.
Abbreviations: GMB, galcanezumab; SE, standard error; LS, least squares.
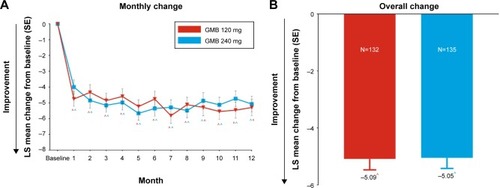
Table S1 Breakdown of responses to the Patient Satisfaction with Medication Questionnaire–Modified by galcanezumab dose group and visit
