Figures & data
Figure 1 Process of identification and selection of studies.
Abbreviations: GDL, guidelines; SR, systematic reviews; SDM, shared decision-making.
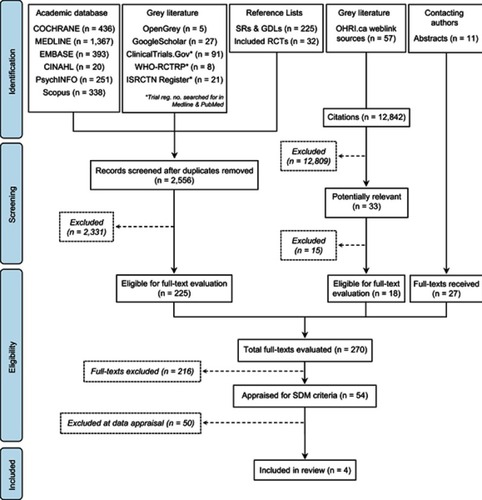
Table 1 Characteristics of included studies
Table 2 Methodological features of included studies
Table 3 Outcome effect estimates for SDM compared to usual care for decisions about prostate cancer treatment
Figure 2 Outcomes reported in the included studies.
Notes: Green = quantitative data; Yellow = qualitative data; Red = no outcome data.
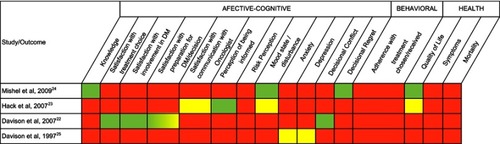
Table S1 PRISMA checklist for the reporting of the systematic review
Table S2 Search strategy for OVID Medline
