Figures & data
Table 1 Designs for studies of budesonide/formoterol pMDI
Table 2 Efficacy of budesonide/formoterol pMDI in patients with asthma
Figure 1 Onset of Effect Questionnaire: Percentage of patients who indicated that they could feel their study medication begin to work right away (A) and that they were satisfied with how quickly they felt their study medication begin to work (B).Citation29 Statistical analyses comparing FM DPI vs BUD pMDI and BUD pMDI + FM DPI vs BUD pMDI and PBO not performed in study I.
Abbreviations: BUD, budesonide; DPI, dry powder inhaler; FM, formoterol; PBO, placebo; pMDI, pressurized metered-dose inhaler.
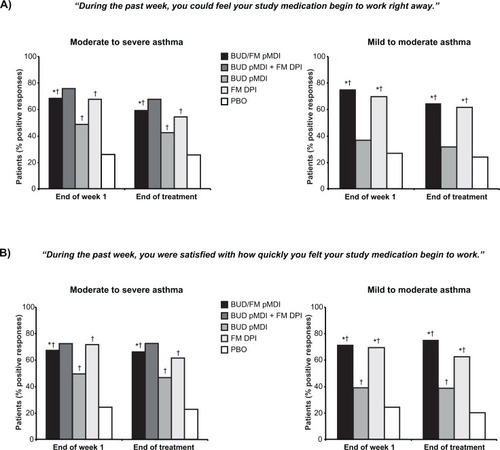
Figure 2 Mean PSAM scores at the end of treatment in patients with (A) moderate to severeCitation31 or (B) mild or moderateCitation32 persistent asthma.
Abbreviations: BUD, budesonide; DPI, dry powder inhaler; FM, formoterol; PBO, placebo; pMDI, pressurized metered-dose inhaler; PSAM, Patient Satisfaction with Asthma Medication.
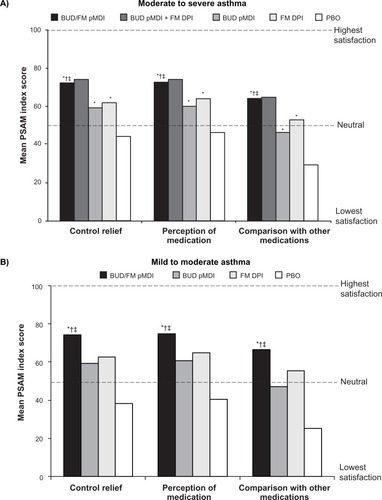
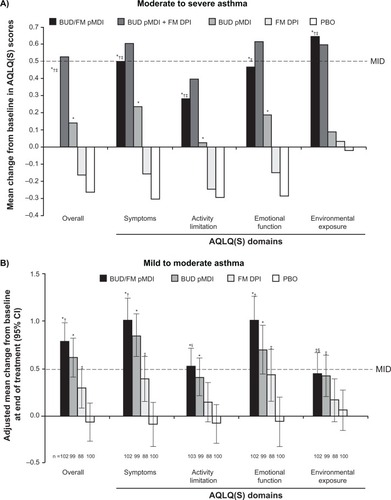
Figure 3 Adjusted mean change from baseline to end of treatment in AQLQ(S) overall and domain scores in patients with (A) moderate to severeCitation31 or (B) mild to moderateCitation32 persistent asthma.
Notes: A: *P < 0.01 vs PBO; †P < 0.05 vs BUD; ‡P < 0.001 vs FM. B: *P < 0.001 vs PBO; †P < 0.001 vs FM; ‡P < 0.05 vs PBO; §P < 0.05 vs FM. Copyright © 2008. Elsevier. Reprinted with permission from Chervinsky P, Baker J, Bensch G, et al. Patient-reported outcomes in adults with moderate to severe asthma after use of budesonide and formoterol administered via 1 pressurized metered-dose inhaler. Ann Allergy Asthma Immunol. 2008;101(5):463–473.Citation31 Copyright © 2008. Informa Healthcare. Murphy K, Nelson H, Parasuraman B, Boggs R, Miller C, O’Dowd L. The effect of budesonide and formoterol in one pressurized metered-dose inhaler on patient-reported outcomes in adults with mild-to-moderate persistent asthma. Curr Med Res Opin. 2008;24(3): 879–894.Citation32