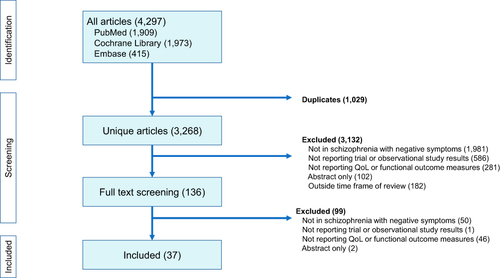
Figures & data
Table 1 Summary of Included Publications
Table 2 Functional Outcome Measures Identified in the Literature Review
Table 3 Quality of Life Measures Identified in the Literature Review