Figures & data
Figure 1 (A) The phylogenetic tree of SARS-like coronaviruses complete genome sequences. (B) The complete genome sequences of SARS-CoV, MERS-CoV, and SARS-CoV-2.
Notes: Reprinted from Journal of Pharmaceutical Analysis, Vol 10/edition number 2, Li X, Geng M, Peng Y, Meng L, Lu S, Molecular immune pathogenesis and diagnosis of COVID-19, Pages No.102–108, Copyright (2020), with permission from Elsevier.Citation8
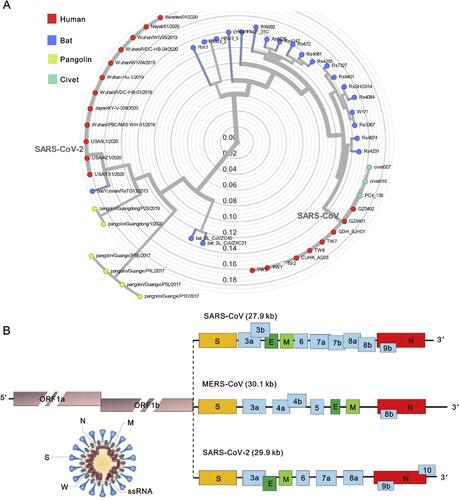
Figure 2 A vision of coronavirus with the minimal set of structural proteins.
Notes: Reprinted from Advances in Virus Research, Vol 66, Masters PS, The molecular biology of coronaviruses, Pages No.193–292, Copyright (2006), with permission from Elsevier.Citation9
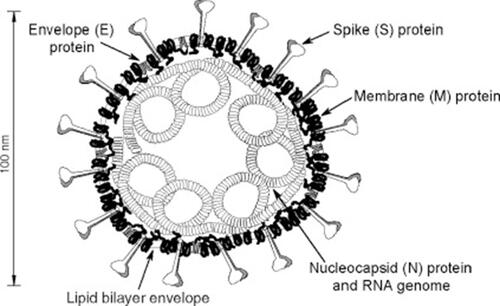
Figure 3 Blocking of S1 Subunit and protease inhibition prevents the SARS-CoV-2 entry into target cell. Therefore S1 subunit and host proteases are potential therapeutic ways for the treatment of COVID-19.
Notes: Reprinted from Archives of Medical Research, Vol 66, Arafah A, Ali S, Yatoo AM, Ali MN, Rehman MU, S1 subunit and host proteases as potential therapeutic avenues for the treatment of COVID-19, In press, Copyright (2020), with permission from Elsevier.Citation10
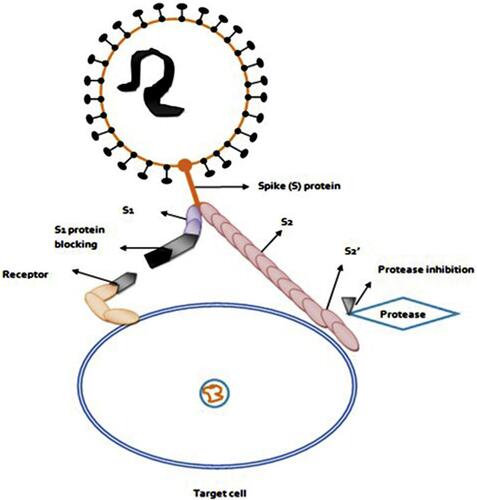
Figure 4 The RBM region of S proteins from SARS-CoV-2 and SARS-CoV alignment.
Notes: (A) The six key amino acids in the S protein interacting with human ACE2 shown by black triangles. (B) Structural Alignment of the ACE2 recognition of RBD from SARS-CoV-2 and SARS-CoV. Human ACE2 (hACE2), SARS-CoV-2 RBD, and SARS-CoV RBD are in orange-red, blue, and green, respectively. Reprinted from Biochemical and Biophysical Research Communications, Vol 526/ Edition 1, Luan J, Lu Y, Jin X, Zhang L, Spike protein recognition of mammalian ACE2 predicts the host range and an optimized ACE2 for SARS-CoV-2 infection, Pages No.165–169, Copyright (2020), with permission from Elsevier.Citation64
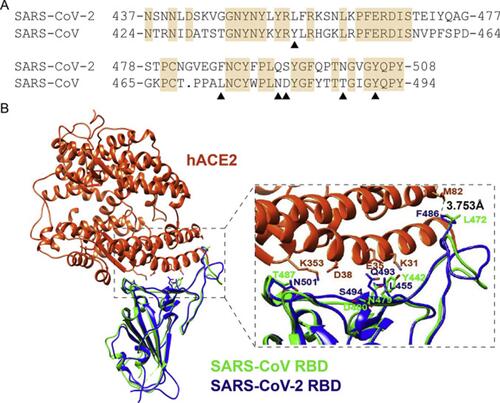
Figure 5 The interaction among nervous, endocrine, and immune systems, modulated by the secreted hormones, neurotransmitters, and cytokines.
Notes: Reproduced from Szałach ŁP, Lisowska KA, Cubała WJ. The influence of antidepres-sants on the immune system. Arch Immunol Ther Exp (Warsz). 2019;67(3):143–151.Citation86
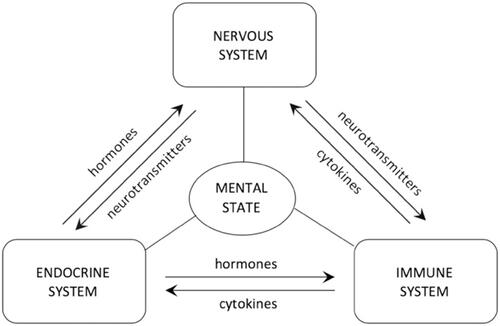
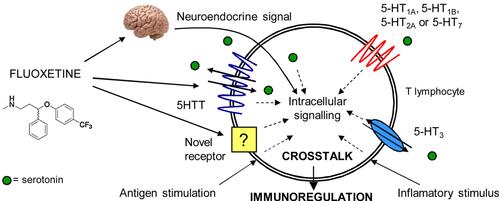
Figure 6 The crosstalk effect of fluoxetine in intracellular signaling and its immune-regulatory role.
Notes: Reprinted from Pharmacological Research, Vol 109, Di Rosso ME, Palumbo ML, Genaro AM, Immunomodulatory effects of fluoxetine: a new potential pharmacological action for a classic antidepressant drug?, Pages No.101–107, Copyright (2016), with permission from Elsevier.Citation87