Figures & data
Figure 1 Vertebral bodies before (A) and after (B) treatment for 14 months for a child with Type I osteogenesis imperfecta.
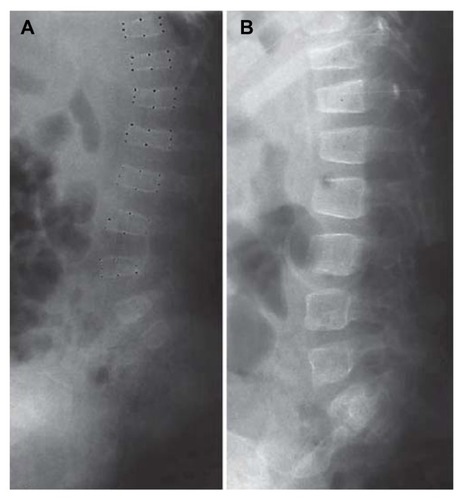
Figure 2 Mean serum alkaline phosphatase levels during the 36-month study for the intramuscular and intravenous neridronate regimens.
Abbreviations: NER, neridronate; i.v., intravenous; i.m., intramuscular.
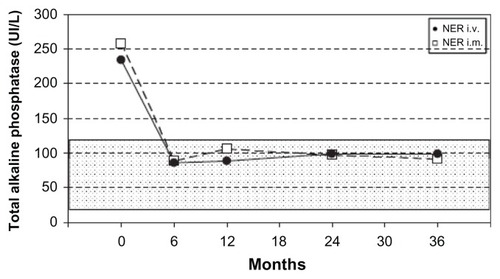
Figure 3 Double-blind phase: VAS trends from baseline to day 40 in patients with complex regional pain syndrome type I treated with neridronate or placebo.
Abbreviations: VAS, visual analog scale; SD, standard deviation.
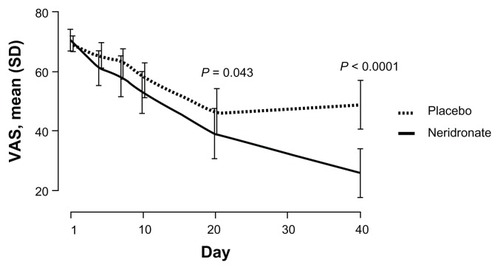
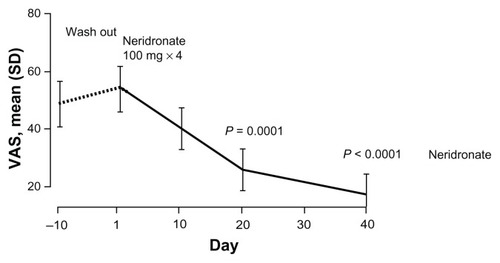
Figure 4 VAS values at the end of the follow-up period of the double-blind phase (day −10) and after the treatment course with intravenous neridronate in patients with complex regional pain syndrome type I.
Abbreviations: VAS, visual analog scale; SD, standard deviation.