Figures & data
Table 1 Demographics and Baseline Characteristics
Figure 1 Device usage by lesion (N=558) assessed by angiographic core lab. *Hawk: Turbohawk, Silverhawk, Hawk One.
Abbreviations: BMS, bare-metal stent; DCB, drug-coated balloon; DES, drug-eluting stent; OAS, orbital atherectomy system; PTA, percutaneous transluminal angioplasty.
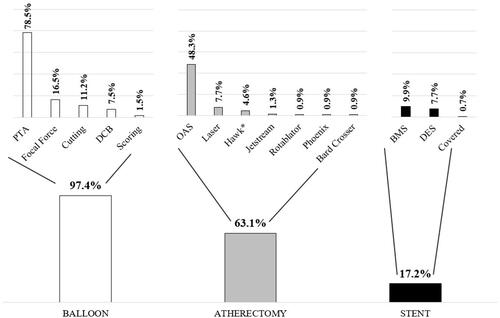
Figure 2 Changes in quality of life from baseline to 12 months (VascuQoL). Vascular Quality of Life Questionnaire: (A) Total score; (B) Physical activity subdomain score; (C) Symptom subdomain score; (D) Pain subdomain score; (E) Emotional subdomain score; and (F) Social subdomain score. Higher subdomain scores indicate better rating of health. Value presented as mean as recorded at each follow-up visit.
Abbreviations: BL, baseline; 30D, 30 days; 6M, 6 months; 12M, 12 months.
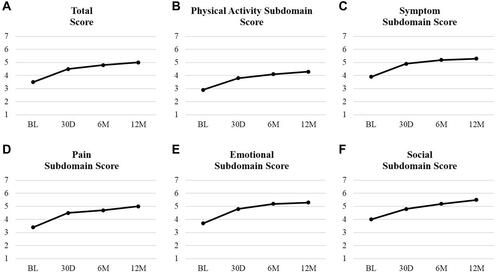
Table 2 Freedom from Major Adverse Events and Amputation Free Survival at 30 Days, 6 Months, and 12 Months
Figure 3 Kaplan–Meier curves for freedom from major adverse events (A) and amputation free survival (B) with number of subjects at risk.
Abbreviation: MAE, major adverse events.
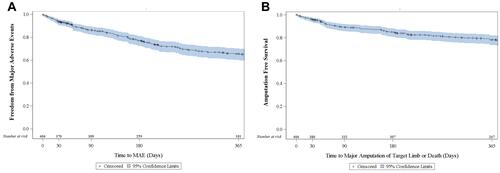
Table 3 Target Limb Wound Healing
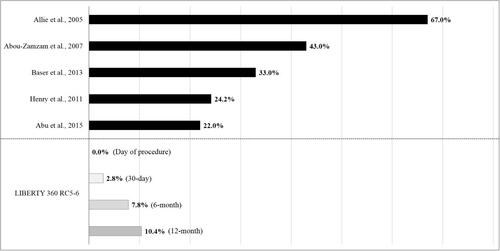
Figure 4 Critical limb ischemia (CLI) patients without revascularization (primary amputation treatment) vs LIBERTY 360 RC5-6 CLI patients (primary endovascular treatment). This summary graph shows the primary amputation rates presented in the literature and the major target limb amputation rates reported in LIBERTY, but it is not a head-to-head comparison since the analyses described vary in design.