Figures & data
Table 1 Pharmacological characteristics of the old and new anticoagulants used for treatment of VTE
Table 2 Clinical trials with DOAC in the treatment of VTE
Figure 1 Recurrent VTE in clinical trials with DOAC in the treatment of VTE.
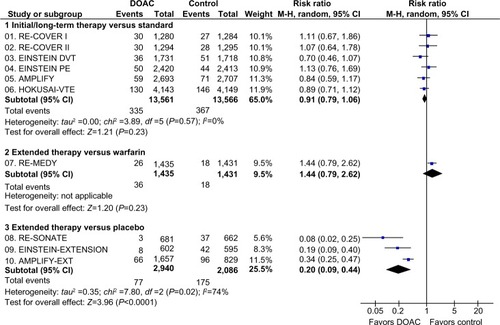
Figure 2 Subgroup analysis of recurrent VTE depending on index event (PE or DVT) in clinical trials with DOAC in the treatment of VTE.
Abbreviations: AMPLIFY, Efficacy and Safety Study of Apixaban for the Treatment of Deep Vein Thrombosis or Pulmonary Embolism; AMPLIFY-EXT, Efficacy and Safety Study of Apixaban for Extended Treatment of Deep Vein; CI, confidence interval; df, degrees of freedom; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; EINSTEIN DVT, Oral Direct Factor Xa Inhibitor Rivaroxaban in Patients With Acute Symptomatic Deep Vein Thrombosis; EINSTEIN-EXTENSION, Once-Daily Oral Direct Factor Xa Inhibitor Rivaroxaban In The Long-Term Prevention Of Recurrent Symptomatic Venous Thromboembolism In Patients With Symptomatic Deep-Vein Thrombosis Or Pulmonary Embolism; EINSTEIN PE, Oral Direct Factor Xa Inhibitor Rivaroxaban in Patients With Acute Symptomatic Pulmonary Embolism; HOKUSAI-VTE, Comparative Investigation of Low Molecular Weight (LMW) Heparin/Edoxaban Tosylate (DU176b) Versus (LMW) Heparin/Warfarin in the Treatment of Symptomatic Deep-Vein Blood Clots and/or Lung Blood Clots; M-H, Mantel–Haenszel; PE, pulmonary embolism; RE-COVER I, Efficacy and Safety of Dabigatran Compared to Warfarin for 6 Month Treatment of Acute Symptomatic Venous Thromboembolism; RE-COVER II, Phase III Study Testing Efficacy and Safety of Oral Dabigatran Etexilate versus Warfarin for 6 Month Treatment for Acute Symptomatic Venous Thromboembolism (VTE); RE-MEDY, Secondary Prevention of Venous Thrombo Embolism (VTE); RE-SONATE, Twice-daily Oral Direct Thrombin Inhibitor Dabigatran Etexilate in the Long Term Prevention of Recurrent Symptomatic VTE; VTE, venous thromboembolism.
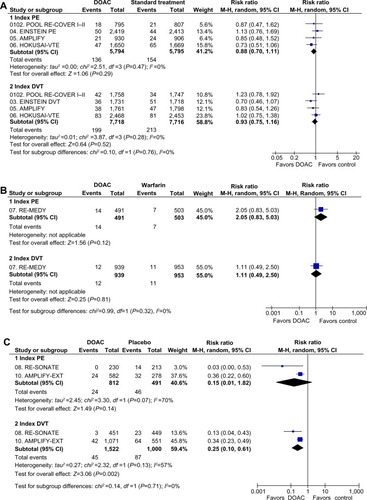
Figure 3 Major and clinically relevant nonmajor bleeding in clinical trials with DOAC in the treatment of VTE.
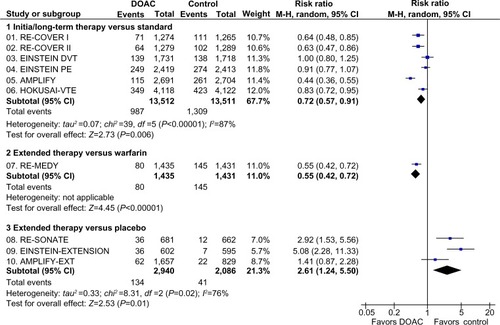
Figure 4 Subgroup analysis of major and clinically relevant nonmajor bleeding events depending on index event (PE or DVT) in clinical trials with DOAC in the treatment of VTE.
Abbreviations: AMPLIFY-EXT, Efficacy and Safety Study of Apixaban for Extended Treatment of Deep Vein; CI, confidence interval; df, degrees of freedom; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; EINSTEIN DVT, Oral Direct Factor Xa Inhibitor Rivaroxaban in Patients With Acute Symptomatic Deep Vein Thrombosis; EINSTEIN PE, Oral Direct Factor Xa Inhibitor Rivaroxaban in Patients With Acute Symptomatic Pulmonary Embolism; HOKUSAI-VTE, Comparative Investigation of Low Molecular Weight (LMW) Heparin/Edoxaban Tosylate (DU176b) Versus (LMW) Heparin/Warfarin in the Treatment of Symptomatic Deep-Vein Blood Clots and/or Lung Blood Clots; M-H, Mantel–Haenszel; PE, pulmonary embolism; VTE, venous thromboembolism.
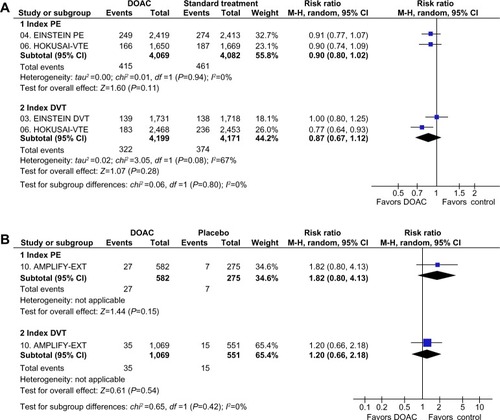
Table 3 Pooled risks of recurrent VTE, CRB, and mortality by index event in patients receiving anticoagulant therapy (DOAC or heparin/warfarin)*
Table 4 Summary of prescribing information of the DOAC in the treatment of VTE
