Figures & data
Figure 1 Serum MPO concentration was measured in a typical ELISA. Five autistic children (A) with GI disease, two autistic children with no GI disease controls (C*), and three nonautistic children with no GI disease controls (C**) were tested. Four replicate samples were tested for each individual.
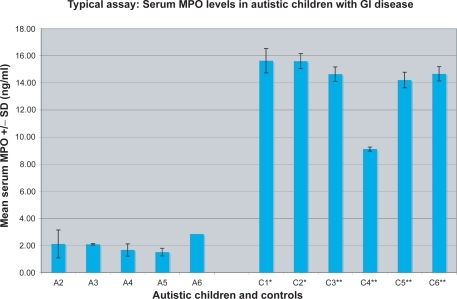
Figure 2 MPO serum concentration was established for each individual by testing and correlating to known standards of various concentrations of MPO (15–1.875 ng/ml) as well as a negative control (serum diluent alone).
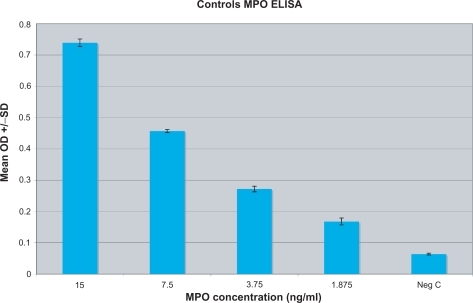
Figure 3 The mean ± SD MPO concentration (ng/ml) of 40 autistic children with chronic digestive disease (most with ileo-colonic LNH and inflammation of the colorectum, small bowel and/or stomach) (autistic GI), and 48 controls (12 age matched autistic children with no GI disease; control autistic, no GI), 20 age-matched siblings of autistic children, without autism or GI disease (control, no autism, no GI), and 16 nonautistic individuals with no family history of autism (control no family history autism).
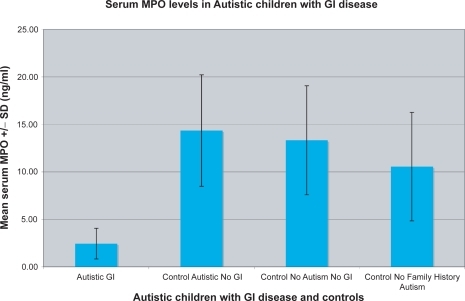
Table 1 The total data from two assays of experimental and control groups is represented. The mean, standard deviation and standard error of the mean of MPO concentration (ng/ml) of 40 autistic children with chronic digestive disease (most with ileo-colonic LNH and inflammation of the colorectum, small bowel and/or stomach) (autistic GI), and 48 controls (12 age-matched autistic children with no GI disease; autistic, no GI), 20 age-matched siblings of autistic children, without autism or GI disease (nonautistic, no GI), and 16 nonautistic individuals with no family history of autism (no family history autism)
Table 2 MPO concentration of autistic children with GI disease with ANCA (anti-MPO or anti-MPO/anti-PR3; bold) and without ANCA are compared (p = 0.3718)
Table 3 MPO concentration of autistic children with GI disease with (bold) and without LNH, with (bold) and without erythema and with (bold) and without severe total GI disease are compared