Figures & data
Table 1 Long-term effect of combination therapy in COPD
Table 2 Absolute and relative risk reductions for exacerbations of COPD requiring medical intervention with number needed to treat in three randomized and double-blind multinational trials
Table 3 Definitions of COPD exacerbation used in combination therapy studies
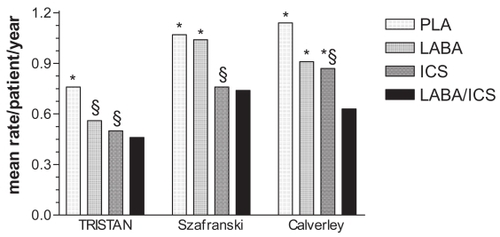
Figure 1 Total severe exacerbation rate.
Notes: PLA, placebo; LABA, salmeterol 50 μg bid or formoterol 12 μg bid; ICS, fluticasone propionate 500 μg bid or budesonide 400 μg bid; LABA/ICS, salmeterol 50 μg/fluticasone 500 μg bid or formoterol 12 μg/budesonide 400 μg bid. * difference between combination treatment and single compound treatment and/or PLA statistically significant (p<0.05); § difference between single compound treatment and PLA statistically significant (p<0.05). Data from CitationCalverley et al 2003a (TRISTAN); CitationCalverley et al 2003b; CitationSzafranski et al 2003.
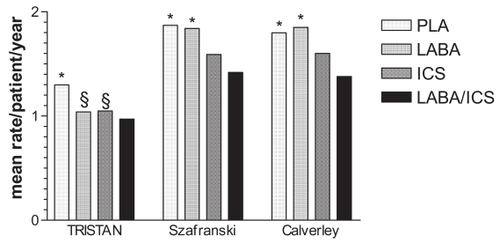
Figure 2 Rate of exacerbations requiring oral corticosteroids.