Figures & data
Table 1 Factors affecting patients’ attitudes to COPD therapy and their preferences for a specific inhaler deviceCitation12–Citation17
Table 2 Items and scoring for the patient satisfaction and preference questionnaire (PASAPQ)Citation44,Citation45
Table 3 Summary of study methods and outcomes of non-comparative trials of Respimat® Soft Mist™ Inhaler (SMI) that assessed patient satisfaction
Table 4 Summary of study methods and satisfaction outcomes of trials that compared Respimat® Soft Mist™ Inhaler (SMI) with another inhaler device using a validated assessment tool (Patient Satisfaction and Preference Questionnaire; PASAPQ) to assess satisfaction scores
Figure 1 Proportion of patients indicating preference for Respimat® Soft Mist™ Inhaler (SMI) or an alternative inhaler device in 3 studies that used the Patient Satisfaction and Preference Questionnaire (PASAPQ): A) Pressurized metered-dose inhaler (pMDI) in a clinical study (n = 224);Citation27 B) Turbuhaler® in a clinical study (n = 153);Citation48 C) Diskus® in an observational study (n = 150).Citation49
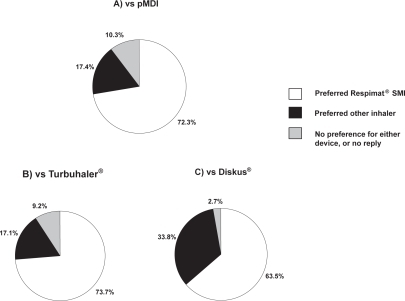
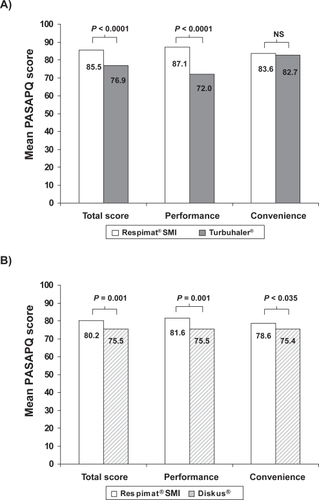
Figure 2 Mean PASAPQ satisfaction scores for Respimat® SMI compared with A) Turbuhaler® in a clinical studyCitation48 and B) Diskus® in an observational study.Citation49 Total scores and performance and convenience domain scores are transformed to a scale of 0 to 100 points; P values for differences between devices are from general linear models analysis (Turbuhaler®) and paired t-test (Diskus®).