Figures & data
Figure 2 Percentage of patients in whom pretreatment isolates that were susceptible to protease inhibitors remained so after failure of darunavir/ritanovir (DRV/RTV) or lopinavir/ritanovir (LPV/RTV) in the TITAN trial. Drawn from data of de Meyer.Citation35
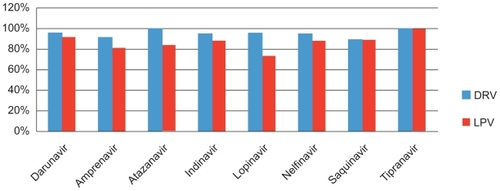
Table 1 Recommended dose for pediatric patients (6 to <18 years of age) for Prezista® tablets with ritonavir body weight doseCitation36
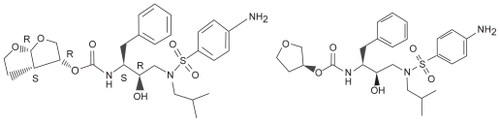
Appendix 1 Interactions with darunavir/ritonavirCitation36,Citation43