Figures & data
Figure 1 The glutamate–NO–cGMP pathway. Activation of ionotropic (mainly NMDA) glutamate receptors leads to increased intracellular calcium (Ca2+) which, after binding to calmodulin (CM), activates neuronal nitric oxide synthase (nNOS) leading to increased production of NO, which in turn activates soluble guanylate cyclase (sGC), resulting in increased formation of cyclic 3′-5′-guanosine monophosphate (cGMP) from guanosine-5′-triphosphate (GTP). Part of the cGMP formed is released to the extracellular space.
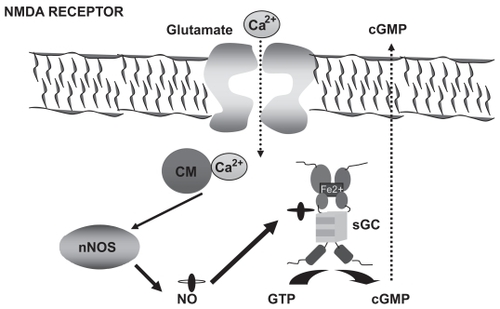
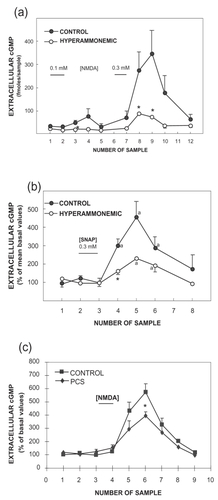
Figure 2 Hyperammonemia impairs the function of the glutamate–NO–cGMP pathway in the cerebellum in vivo by reducing NO-induced activation of soluble guanylate cyclase. The effects of NMDA or SNAP on extracellular cGMP in the cerebellum of control, hyperammonemic rats without liver failure (2a and 2b) and with chronic liver failure (2c) were analyzed by in vivo microdialysis in freely moving rats. NMDA (0.1 and 0.3 mM) or SNAP (0.3 mM) was administered in the perfusion stream for 20 minutes at the times indicated by the horizontal bars. Perfusion was carried out at 3 μL/minute, samples were collected every 20 minutes, and cGMP was determined. Data are presented as a percentage of basal values. Data were analysed using two-way ANOVA.
a intragroup comparison values significantly different from baseline (p < 0.05 by Kruskal-Wallis test with post hoc Dunnett’s test; * values significantly different from control (p < 0.05).
Figure 2a and 2b from: CitationHermenegildo C, Montoliu C, Llansola M, et al. 1998. Chronic hyperammonemia impairs glutamatenitric oxide-cyclic GMP pathway in cerebellar neurons in culture and in the rat in vivo. Eur J Neurosci, 10:3201–9;
Figure 2c from: CitationMonfort P, Corbalán R, Martínez L, et al. 2001. Altered content and modulation of soluble guanylate cyclase in the cerebellum of rats with portacaval anastomosis. Neuroscience, 104:1119–25.