Figures & data
Table 1 Safety and Tolerability Trial I (CitationKeck et al 2003)
Table 2 Safety and Tolerability Trial II (CitationPotkin et al 2005)
Figure 1 Mean decrease in MRS pure mania vs mixed episodes after 21 days (estimated from CitationPotkin et al 2004).
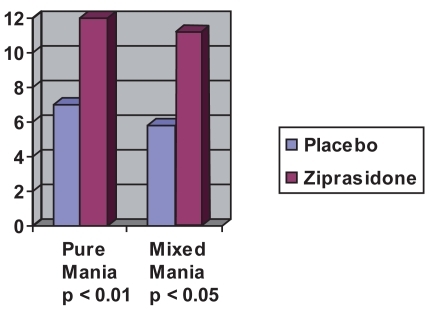
Figure 2 Decrease in MRS psychotic mania vs non-psychotic mania after 21 days (estimated from CitationPotkin et al 2004).
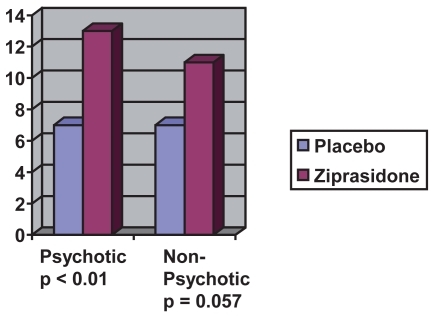
Table 3 Ziprasidone augmentation with lithium (CitationWeisler et al 2003)
Table 4 Adjunctive ziprasidone with lithium 52-week open label extension (CitationWeisler et al 2004)
Table 5 Ziprasidone 52-week open label extension (CitationKeck et al 2004)