Figures & data
Figure 1 Key molecular pathways in renal cell carcinoma pathogenesis and the points at which the currently approved agents function.
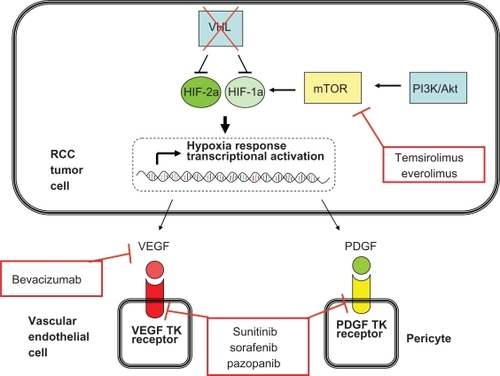
Table 1 Kinase inhibitory concentrations (IC50, nmol) for multitargeted tyrosine kinase inhibitors for renal cell carcinoma (includes tivozanib and axitinib which are in Phase III study, but not yet approved) arranged in order of VEGFR2 potency
Table 2 Toxicities of pazopanib from the Phase III study in renal cell carcinoma (n = 290) which occurred at a rate of 10% or more. The toxicities are arranged in order of the most common adverse (any grade) to the least common. Grade 3/4 toxicities with frequency of occurrence are listed as well
Table 3 Results from Phase III trials of currently approved agents for the treatment of renal cell carcinoma
Table 4 Level 1 evidence for use of molecularly targeted agents in renal cell carcinoma showing potential treatment settings and agents which have a high level of clinical evidence supporting their use