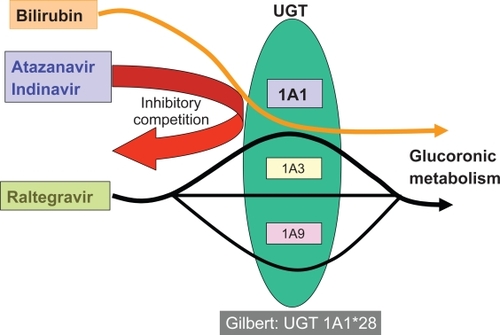
Figures & data
Table 1 Different genotypic resistance scores for atazanavir (boosted or unboosted) in relation to clinical responses
Table 2 Pharmacokinetic parameters at steady state after atazanavir 400 mg once daily and after atazanavir 300 mg with ritonavir 100 mg once daily with a light meal in HIV-infected patients
Table 3 Main drug interactions with atazanavir 400 mg or atazanavir/ritonavir 300/100 mg/day
Table 4 Main randomized studies with atazanavir in antiretroviral-naïve patients
Table 5 Main randomized studies with atazanavir in antiretroviral-experienced patients with virologic failure
Table 6 Main switch studies with atazanavir