Figures & data
Figure 1 Hydrolysis of piperacillin followed by formation of a piperacillin dimer with low solubility.
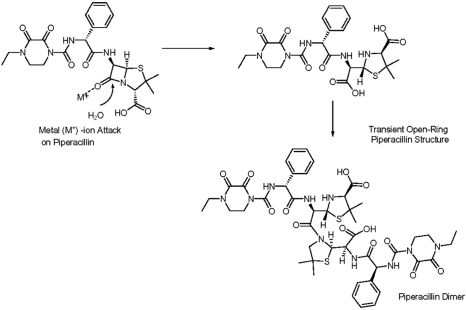
Table 1 Combination matrix for reformulated Tazocin® plus gentamicin (reconstituted and diluted in D2O) at clinically relevant bracketed ranges of concentrations
Table 2 Summary of drug potency results for admixtures of reformulated Zosyn® in various diluents and stored at room temperature for up to 24 hours
Table 3 Summary of drug potency results for simulated Y-site coadministration of reformulated Zosyn® with amikacin in compound sodium lactate intravenous infusion BP (Hartmann’s solution) at room temperature
Table 4 Degradation products of reformulated Zosyn® drug components in the presence of amikacin up to 4 hours at room temperature in compound sodium lactate intravenous infusion BP (Hartmann’s solution)
Table 5 Simulated Y-site coadministration of reformulated Zosyn® with amikacin in different admixture diluents (Potency of antibiotics at 4 hours)
Table 6 Reformulated Tazocin® : Simulated Y-site compatibility by NMR for Ratiopharm® German gentamicin drug product
Table 7 Summary of subvisible particulate counts by HIAC for simulated Y-site coadministration of reformulated Zosyn® and amikacin
Table 8 Summary of subvisible particulate counts by HIAC for simulated Y-site coadministration of reformulated Zosyn® and gentamicin