Figures & data
Figure 1 The linear pathway leading from insulin deficiency, through hyperglycemia to diabetes complications.
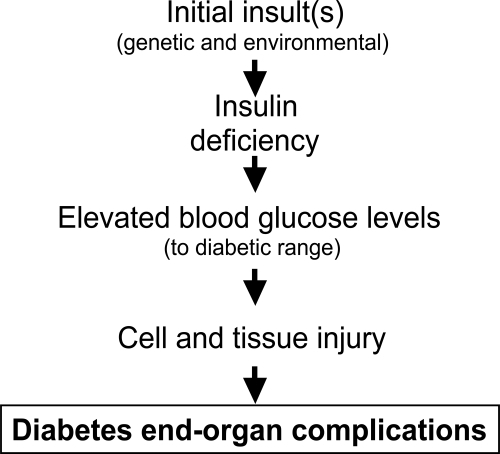
Figure 2 Schematic diagram indicating how hemodynamic and metabolic factors, and growth factors, can network to cause tissue damage. Inflammation and fibrosis occur variably in tissue at different stages of diabetes complications.
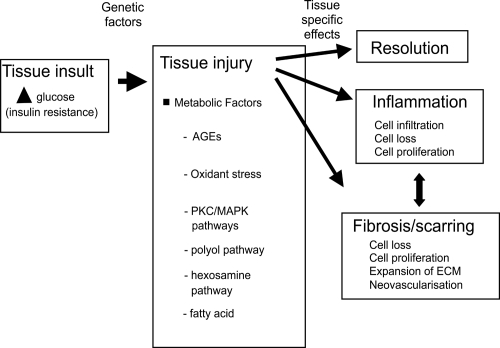
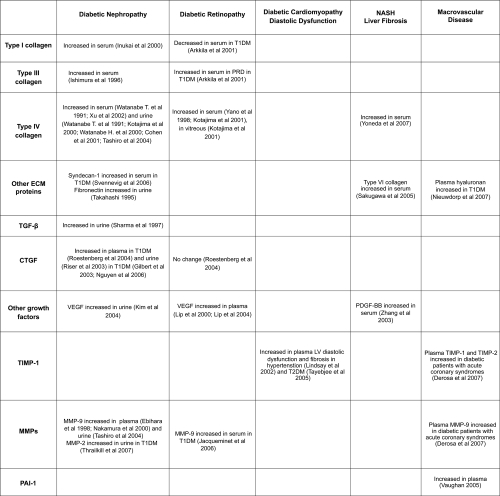
Figure 3 The major growth factors implicated in diabetes complications. The prosclerotic ones involved in human diabetic fibrosis are currently thought to be TGF-β and CTGF.
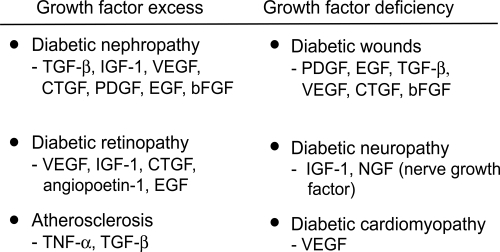
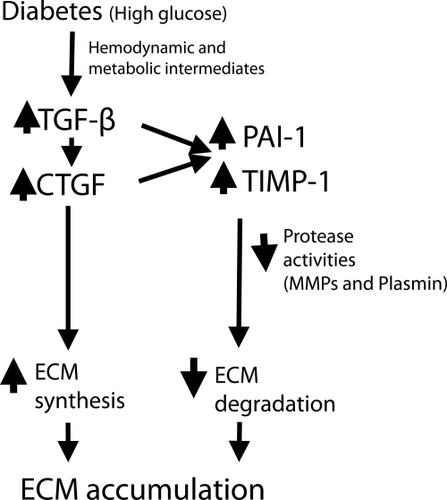
Figure 4 One pathogenic pathway by which high glucose in diabetes and hypertension work through prosclerotic growth factors to dysregulate ECM turnover. Both TGF-β and CTGF have been shown to induce TIMP-1 and PAI-1, resulting in reduced MMP and plasmin activity. This paradigm best applies to diabetic renal disease.