Figures & data
Table 1 Compelling indications and contraindications in the use of antihypertensive drug classesCitation17
Figure 2 Four out of 6 recommended dual antihypertensive combination therapies include blockers of the renin–angiotensin system.
Reproduced with permission from Mancia G, De Backer G, Dominiczak A, et al. Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2007; 25(6):1105–1187.Citation17 Copyright © Lippincott Williams & Wilkins.
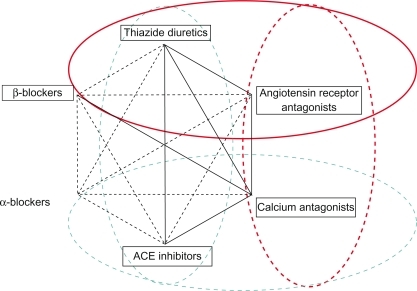
Figure 3 Combination therapy as an escalation option and as first-line therapy.
Reproduced with permission from Mancia G, De Backer G, Dominiczak A, et al. Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2007; 25(6):1105–1187.Citation17 Copyright © Lippincott Williams & Wilkins.
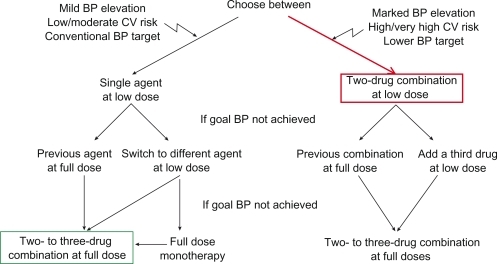
Figure 4 Cost savings in the UK by intensifying antihypertensive drug treatment.Citation15
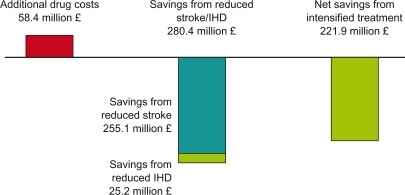
Figure 5 Blood pressure reduction with 32 mg candesartan alone or in combination with 12.5 or 25 mg HCTZ in patients not sufficiently controlled on monotherapy.Citation42
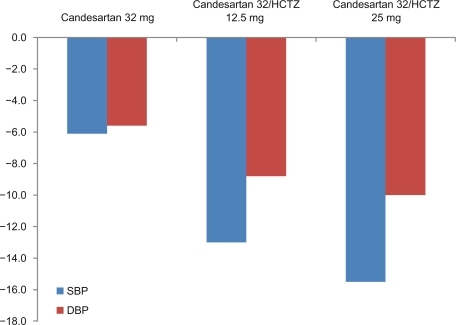
Table 2 Laboratory values at baseline, and mean change (±SD) from baselineTable Footnotea after 8 weeks of treatmentCitation42
Figure 6 Results of the CHARM trial program.Citation52–Citation55
Reprinted from Pfeffer MA, Swedberg K, Granger CB, et al. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. The Lancet. 362:759–766.Citation52 Copyright © 2003, with permission from Elsevier.
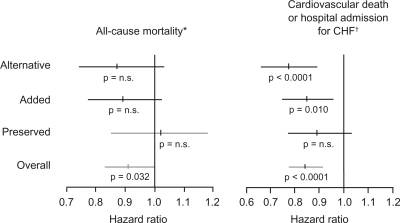
Figure 7 Development of diabetes – results of a meta-analysis.
Reprinted from Lam SK, Owen A. Incident diabetes in clinical trials of antihypertensive drugs. The Lancet. 369:1513–1514.Citation59 Copyright © 2007, with permission from Elsevier.
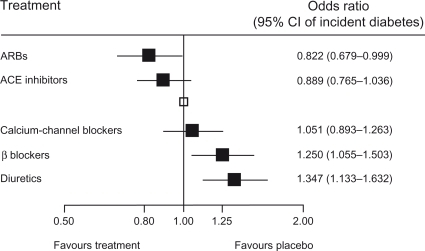
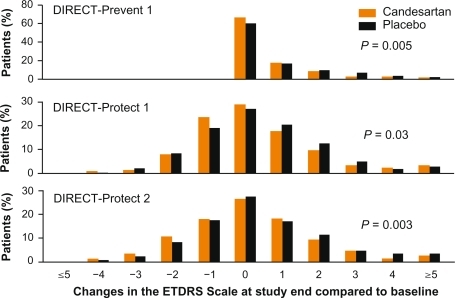
Figure 8 Results of the DIRECT trial program.Citation47,Citation73