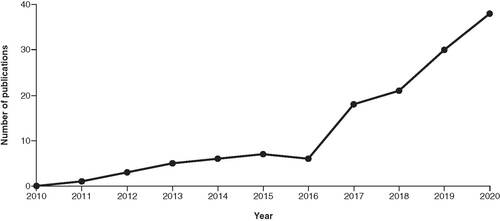
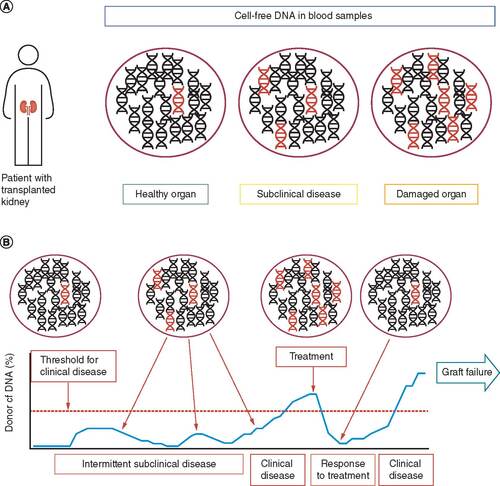
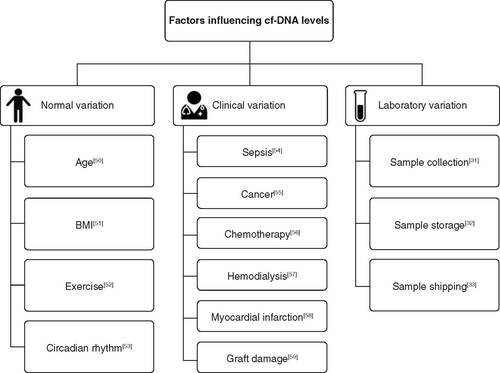
Figures & data
Table 1. Detection cell-free DNA in solid-organ transplant recipients.
Table 2. Commercial testing for cell-free DNA in solid-organ transplant recipients.
Table 3. Results of donor-derived cell-free DNA test from a single commercial provider.