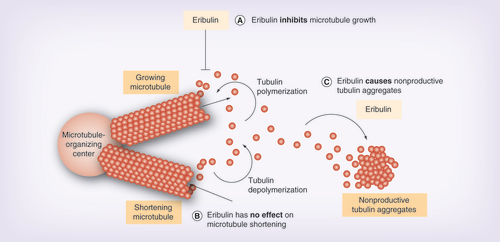
Figures & data
Table 1.
Published Phase II trials for eribulin as monotherapy in advanced breast cancers.
Table 2.
Summary of Phase II results: eribulin as first-line treatment in patients with previously untreated advanced HER+ and HER- breast cancer.
Table 3.
Phase II neurotoxicity profile of eribulin.
Table 4.
Incidence of neuropathy and sensitivity analysis of neuropathy based on a narrow definition of peripheral neuropathy by Common Terminology Criteria for Adverse Events grade.
Table 5.
EMBRACE progression-free survival and best overall tumor response by independent review.
Table 6.
Eribulin versus capecitabine: response rates.
![Figure 2. Kaplan–Meier of EMBRACE overall survival. TPC: Treatment of physician’s choice.Reproduced with permission from [Citation9] © Elsevier (2011).](/cms/asset/19c7ef38-e6af-4de4-8513-0ea273f1507a/ibmt_a_12322894_f0002.jpg)
![Figure 3. Eribulin versus capecitabine. (A) OS. (B) OS in HER2- subgroup. Data are for the intent-to-treat population.*HR Cox model including geographic region and HER2 status as strata. †p-value from stratified log-rank test based on clinical database.HR: Hazard ratio; OS: Overall survival. (A) Reproduced with permission from [Citation33]; (B) Reproduced with permission from [Citation34].](/cms/asset/18c9156d-09bc-40e4-b801-3ce015cde810/ibmt_a_12322894_f0003.jpg)
![Figure 4. Effect of eribulin treatment modification on duration of therapy: type and timing of dose modifications. Reproduced with permission from [Citation41].](/cms/asset/241be88d-4306-4c12-8899-b56097c01c0e/ibmt_a_12322894_f0004.jpg)
