Figures & data
Table 1.
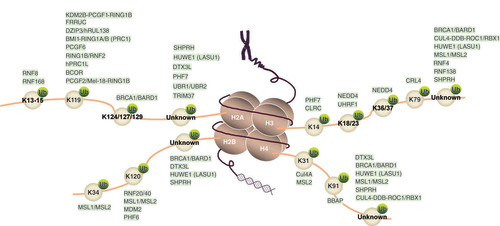
RING finger proteins ubiquitinate different lysine sites of core histones.
Table 2.
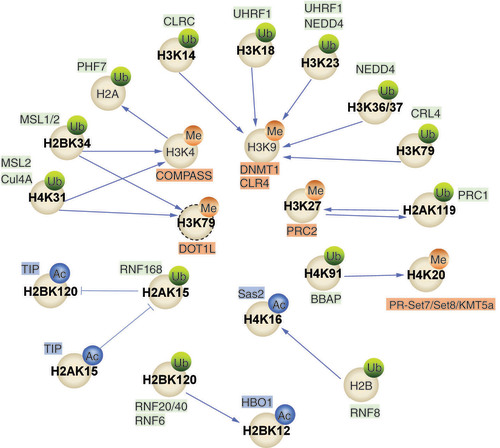
RING finger proteins mediate the crosstalk between histone ubiquitination and histone methylation; the relationship between the interactions is also shown.
Table 3.
RING finger proteins that mediate the crosstalk between histone ubiquitination and histone acetylation; the relationship between the interactions is also listed.