Figures & data
DLBCL: Diffuse large B-cell lymphoma; FL: Follicular lymphoma; MCL: Mantle cell lymphoma; MZL: Marginal zone lymphoma; NHL: Non-Hodgkin lymphoma.
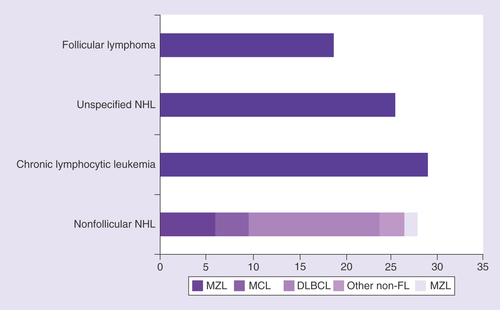
Table 1. Patient demographics.
BR: Bendamustine plus rituximab; FCR: Fludarabine plus cyclophosphamide and rituximab; n/s: Not specified; R-CHOP: Rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisolone; SC: Subcutaneous.
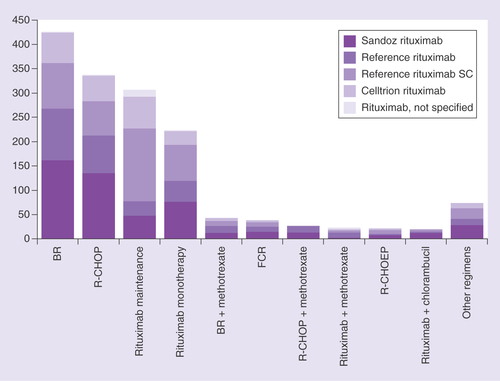
Table 2. Most frequent rituximab protocols identified in the Onkotrakt database.
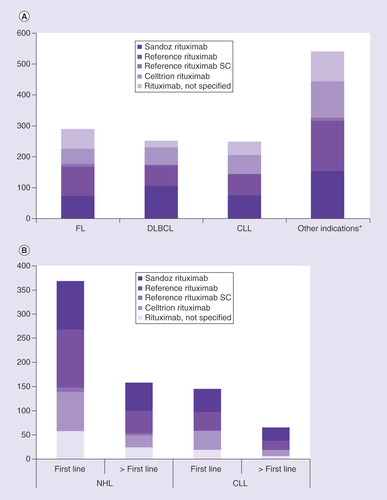
(A) indication and (B) line of therapy.
*Marginal zone lymphoma, mantle cell lymphoma, ‘other nonfollicular lymphoma’, NK/T cell lymphoma, ‘unspecified NHL’.
CLL: Chronic lymphocytic leukemia; DLBCL: Diffuse large B-cell lymphoma; FL: Follicular lymphoma; NHL: Non-Hodgkin lymphoma; NK/T cell: Natural killer/T cell; SC: Subcutaneous.
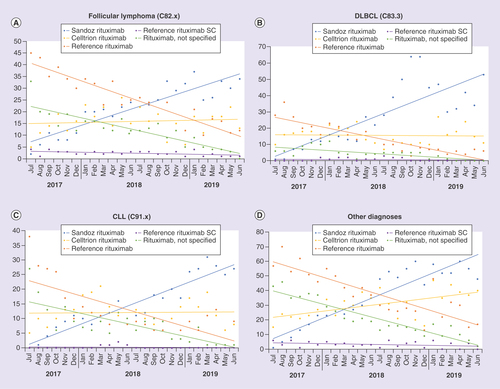
CLL: Chronic lymphocytic leukemia; DLBCL: Diffuse large B-cell lymphoma; SC: Subcutaneous.
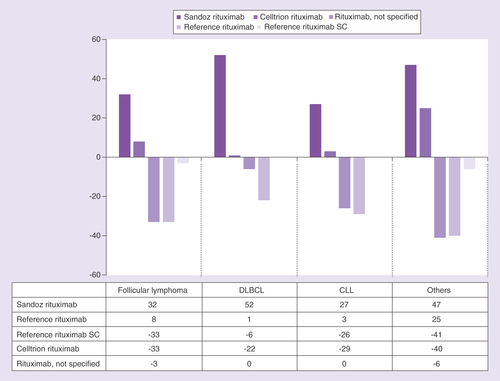
SC: Subcutaneous.
(A) FL, (B) DLBCL, (C) CLL, (D) Other (unspecified non-Follicular lymphoma, Marginal zone lymphoma, Mantle cell lymphoma, Natural killer/T cell cell lymphoma). Data presented as polynomial trend lines of the percentage prescriptions per month.
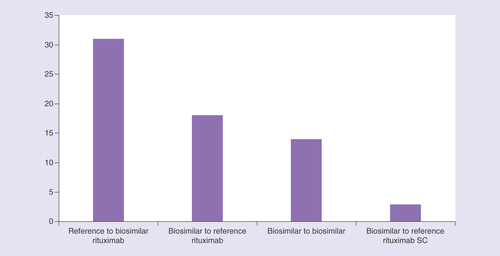
CLL: Chronic lymphocytic leukemia; DLBCL: Diffuse large B-cell lymphoma; NHL; SC: Subcutaneous.