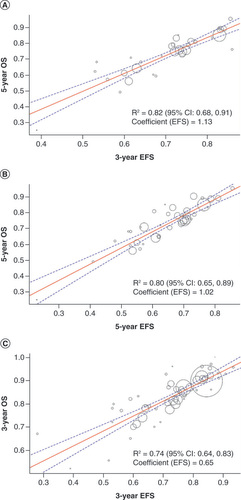
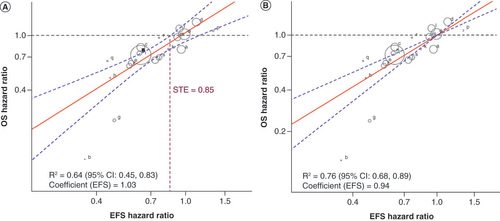
Figures & data
Table 1. Key characteristics of studies included in the base case analysis of individual-level association.
Table 2. Key characteristics of randomized controlled trials included in the base case analysis of individual-level and trial-level associations.