Figures & data
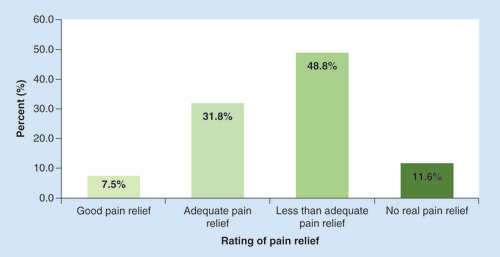
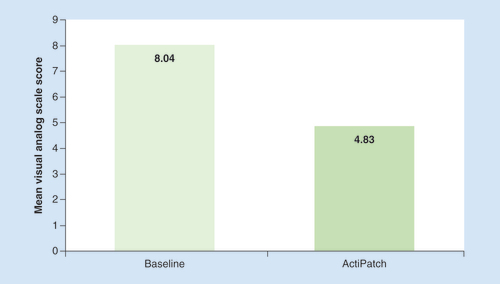
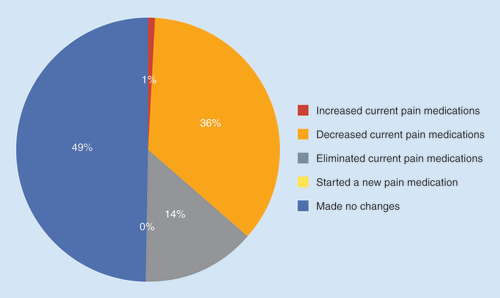
The mean number of adverse effects was 4.6 per subject.
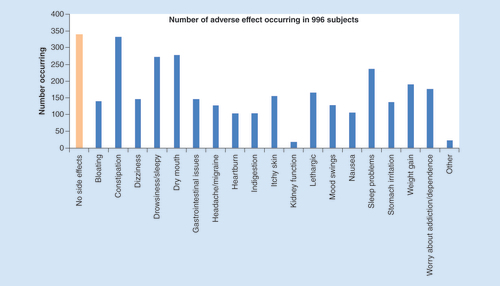
Table 1. Gender distribution.
Table 2. Duration of pain.
Table 3. The reported etiologies show a heterogeneous pain population.
Table 4. Medication use of the subjects in the study.
Table 5. The percent of individuals who reported a 40% or greater visual analog scale scores reduction was 726/1394 or 52%.
Table 6. The intent to continue therapy.
Table 7. Pain duration, analgesic use, percent subjects reporting adverse effects, mean adverse effects and their impact on patients’ quality of life.
Table 8. Subject age group, analgesic use, percent of subjects reporting adverse effects, mean adverse effects and their impact on patients’ quality of life.
Supplemental material
