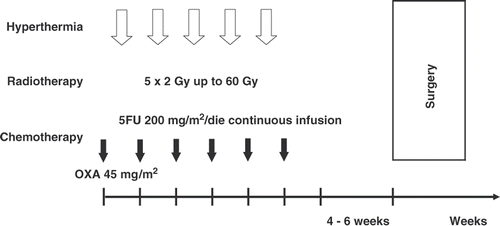
Figures & data
Table I. Patient characteristics (n = 76).
Table II. HT toxicity score, from B. Rau et al. Citation[29].
Table III. CRT–related acute toxicity according to RTOG–EORTC scoring criteria Citation[35], observed in all patients (n = 76).
Table IV. Clinical stage at diagnosis and subsequent pathological stage after neo–adjuvant HT plus CRT and surgery for all patients (n = 76).
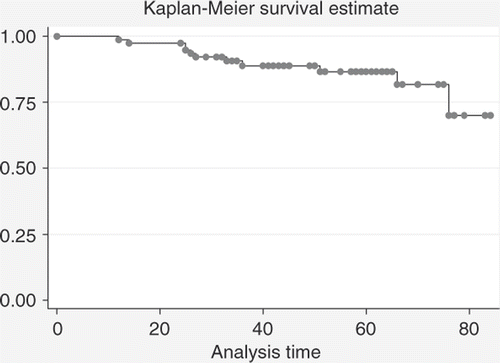
Figure 5. Comparison between DFS estimates for patients with ypN0 status (n = 53) and patients with ypN+ status (n = 23) (log-rank test: p = 0.0008).
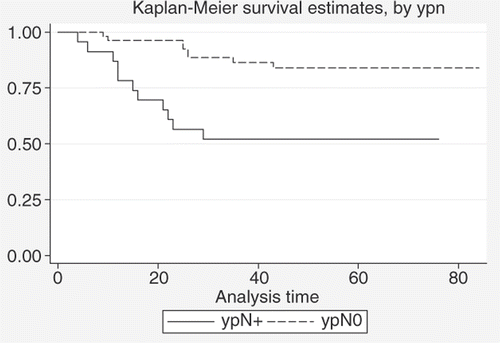
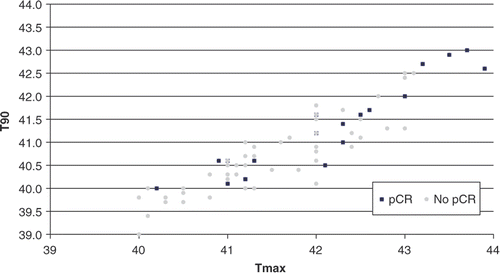
Figure 6. Comparison between DFS estimates for patients with pCR (n = 18) and patients with no pCR (n = 58) after neoadjuvant treatment (log-rank test: p = 0.03).
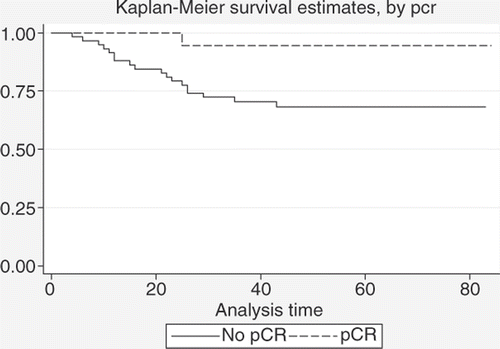
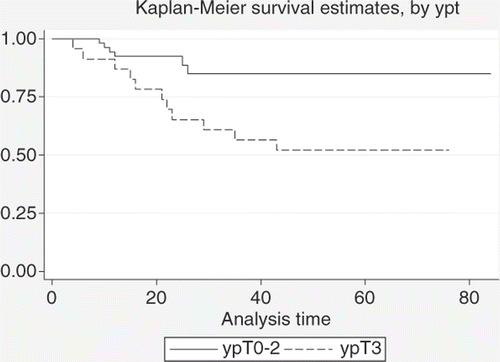
Figure 7. Comparison between DFS estimates for patients with ypT0-2 status (n = 53) and patients with ypT3 status (n = 23) after neoadjuvant treatment (log-rank test: p = 0.002).