Figures & data
Figure 1. Temperature of a cell pellet by induction heating. Cell pellets filling the intercellular space with five different concentrations of ferucarbotran were applied for an AMF. The equilibrium temperatures; 38.3°, 46.5°, 51.6°, 64.3°, and 69.7°C were obtained for the five different concentrations of 20♦, 27▾, 40▴, 60•, and 80▪ mg γ-Fe2O3/mL initial ferucarbotran concentration, respectively (A). The value between the equilibrium temperature and applied heat dose was plotted for three different cancer cell lines (SUIT-2▪, BxPC-3•, and AsPC-1▴) (B). Heat dose of 0.7, 1.5, 2.1, 7.3, and 9.5 W/gtumour, resulted in equilibrium temperatures of 38.3°, 46.5°, 51.6°, 64.3°, and 69.7°C, respectively.
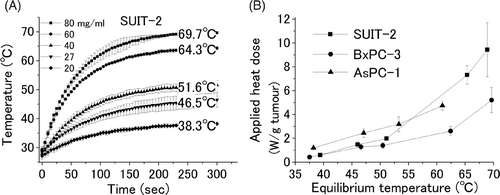
Figure 2. Computer simulation of thermal gradient in a small in vitro cell pellet surrounded by air. Thermal gradients of the 20 µL cell pellet, which was heated with five different heat doses (0.7, 1.5, 2.1, 7.3, and 9.5 W/gtumour) for 300 s are shown (A). The time-dependent change in the temperature at the centre (B) and periphery of pellet (C). Equilibrium temperatures at the centre of the tumour on administration of the above-mentioned heat doses were 38.9°, 43.4°, 51.4°, 62.7°, and 73.3°C, respectively. Those at the periphery were 36.0°, 39.5°, 45.7°, 54.5°, and 62.5°C, respectively.
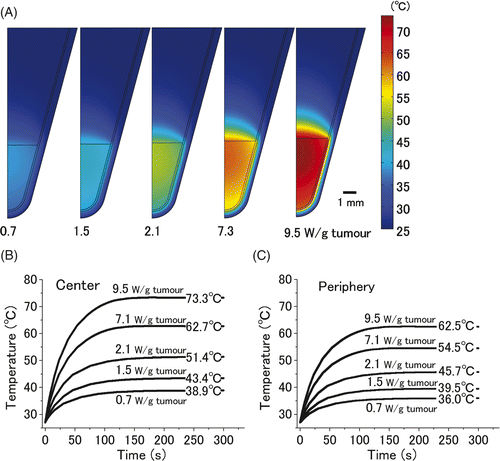
Figure 3. Cytotoxic effect of heat, generated by induction heating. An AMF (100–150 Oe, 114 kHz) was applied to three different cell pellets (SUIT-2, BxPC-3, and AsPC-1) containing ferucarbotran. After 10-min exposure to various temperatures (39°C, 42°C, 46°C, 48°C, 51°C, and 60–65°C), each cell pellet was cultured for 7 days. The numbers of viable cells 1, 3, and 7 days after incubation were counted. The values and bars are the mean and SD of three independent experiments.
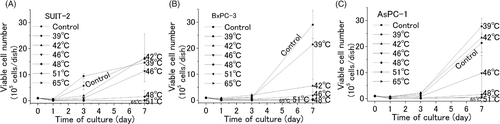
Figure 4. Computer simulation of the thermal gradient in in vivo liver tumour of different sizes. The temperature changes in the 10 mm (A, C) and 5 mm tumours (B, D) with a heat dose of 1.7 W/gtumour for 300 s were simulated. Images of the entire liver and an enlarged image of a part of the tumours (A, B). Time-dependent change in the temperatures at the centre and periphery of the 10 mm (C) and 5 mm (D) tumours. In the simulation of the 10-mm-diameter tumour, the centre and periphery of the 10-mm tumour were heated to 61°C and 50°C, respectively, however, those of the 5-mm tumour were heated only to 44°C and 41°C, respectively.
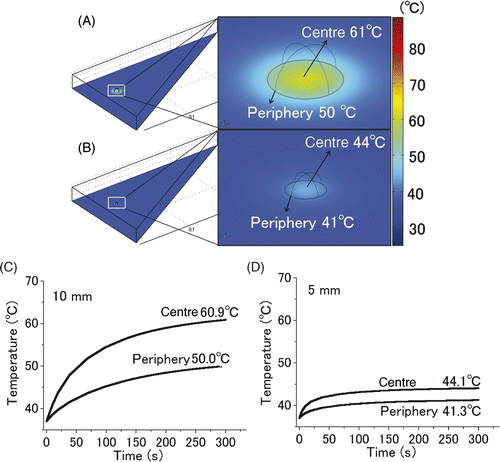
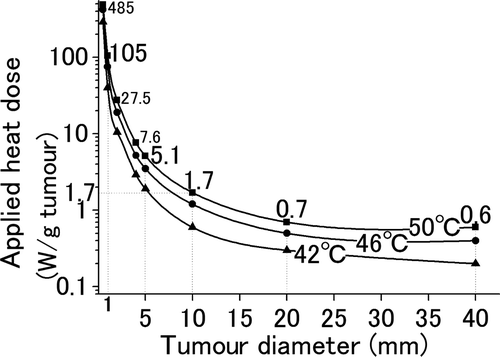
Figure 5. Required heat dose for liver tumours with various diameters. The relationship between the tumour size and the heat dose administered for 300 s to heat the tumours to 42°C (▴), 46°C (•), and 50°C (▪) at their periphery was simulated. In order to increase the temperature of the tumour periphery up to 50°C, tumours with diameters 40, 20, 10, 5, and 1 mm require heat doses of 0.6, 0.7, 1.7, 5.1, and 105 W/gtumour, respectively.