Figures & data
Figure 1. T cell-based immunotherapies share a common requirement for trafficking across vascular checkpoints in tumour tissues. The direct tumouricidal activity of therapeutic interventions including dendritic cell (DC) vaccination, adoptive T cell transfer, and administration of cytotoxic T-lymphocyte antigen-4 (CTLA-4) inhibitors hinge on the ability of cytotoxic CD8+ T cells to traffic across vascular barriers (blue cells in inset) within the tumour microenvironment.
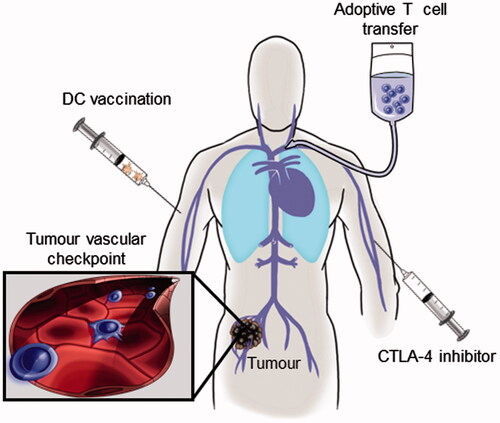
Figure 2. Thermal therapy primes tumour microvessels to support E/P-selectin-dependent and ICAM-1-dependent trafficking of CD8+ effector T cells. (A) Multistep adhesive interactions direct circulating T cells into tumour sites where they can kill tumour cell targets; thermally responsive adhesion events are marked by red asterisks (*). (B) STT treatment of B16-OVA tumours increased intravascular expression of ICAM-1 (red) over baseline normothermal control (NT). The landmark position of vessels in photomicrographs is shown by immunostaining for the CD31 pan-endothelial adhesion molecule (green). Histograms represent quantitative image analysis of relative immunofluorescence staining intensity of CD31 or ICAM-1 on all CD31+ vessels; studies were performed as described by Fisher et al. [Citation35]. Bar, 50 μm.
![Figure 2. Thermal therapy primes tumour microvessels to support E/P-selectin-dependent and ICAM-1-dependent trafficking of CD8+ effector T cells. (A) Multistep adhesive interactions direct circulating T cells into tumour sites where they can kill tumour cell targets; thermally responsive adhesion events are marked by red asterisks (*). (B) STT treatment of B16-OVA tumours increased intravascular expression of ICAM-1 (red) over baseline normothermal control (NT). The landmark position of vessels in photomicrographs is shown by immunostaining for the CD31 pan-endothelial adhesion molecule (green). Histograms represent quantitative image analysis of relative immunofluorescence staining intensity of CD31 or ICAM-1 on all CD31+ vessels; studies were performed as described by Fisher et al. [Citation35]. Bar, 50 μm.](/cms/asset/3cd8f32e-58e4-49e4-82ca-dfa3b68c0394/ihyt_a_807440_f0002_b.jpg)
Figure 3. Thermal therapy induces CD8+ T cell trafficking and target cell apoptosis in the tumour microenvironment. (A) Schematic for combination systemic thermal therapy (STT) and adoptive cell transfer (ACT) of tumour-specific effector CD8+ T cells. Experimental endpoints for analysis of CD8+ T cell homing and TUNEL staining of apoptotic cells in tumour cryosections as well as tumour growth control are described in a recent report [Citation35]. (B) Administration of IL-6–neutralising monoclonal antibody (mAb) 30 min prior to STT blocked induction of CD8+ T cell trafficking (red) and apoptosis of intratumoural cellular targets (green). Relative values for normothermal (NT) mice are shown; nucleated cells are labelled with DAPI (blue). Left, representative photomicrographs of tumour cryosections. Bar, 50 μm. Right, quantification of intratumoural CD8+ T cells and TUNEL+ apoptotic cells per unit area of tumour tissue. *p < 0.001. (C) Thermal induction of ICAM-1-dependent CD8+ T cell trafficking in tumour vessels involves an IL-6 trans-signalling mechanism whereby intracellular JAK-1/2 and STAT3 activation occurs downstream of ligation of an agonistic, soluble form of the IL-6 receptor α binding subunit (sIL-6Rα) and the gp130 signal transducing subunit.
![Figure 3. Thermal therapy induces CD8+ T cell trafficking and target cell apoptosis in the tumour microenvironment. (A) Schematic for combination systemic thermal therapy (STT) and adoptive cell transfer (ACT) of tumour-specific effector CD8+ T cells. Experimental endpoints for analysis of CD8+ T cell homing and TUNEL staining of apoptotic cells in tumour cryosections as well as tumour growth control are described in a recent report [Citation35]. (B) Administration of IL-6–neutralising monoclonal antibody (mAb) 30 min prior to STT blocked induction of CD8+ T cell trafficking (red) and apoptosis of intratumoural cellular targets (green). Relative values for normothermal (NT) mice are shown; nucleated cells are labelled with DAPI (blue). Left, representative photomicrographs of tumour cryosections. Bar, 50 μm. Right, quantification of intratumoural CD8+ T cells and TUNEL+ apoptotic cells per unit area of tumour tissue. *p < 0.001. (C) Thermal induction of ICAM-1-dependent CD8+ T cell trafficking in tumour vessels involves an IL-6 trans-signalling mechanism whereby intracellular JAK-1/2 and STAT3 activation occurs downstream of ligation of an agonistic, soluble form of the IL-6 receptor α binding subunit (sIL-6Rα) and the gp130 signal transducing subunit.](/cms/asset/d0cdf926-ed38-45b2-81b8-8407f57e4b26/ihyt_a_807440_f0003_b.jpg)
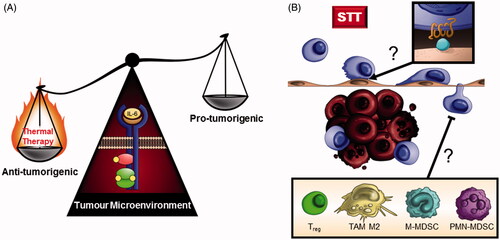
Figure 4. Collaborative intersection between thermal therapy and IL-6 in the tumour microenvironment. (A) Thermal therapy tips the balance of IL-6 activity in the tumour microenvironment from a tumour-promoting inflammatory role to an anti-tumourigenic role supporting T cell-mediated tumour immunity. (B) Outstanding questions remain regarding which chemokine/chemokine receptor interactions (inset) mediate CD8+ T cell trafficking at the vascular interface during heat therapy and whether clinical responses to preconditioning regimens are predicated on the presence of immunosuppressive cell subsets (inset) within the tumour microenvironment.